For each of the following molecules, complete the Lewis structure and use the VSEPR model to determine
Question:
For each of the following molecules, complete the Lewis structure and use the VSEPR model to determine the bond angles around each central atom. Note that the drawings are only skeleton structures and may depict the angles incorrectly.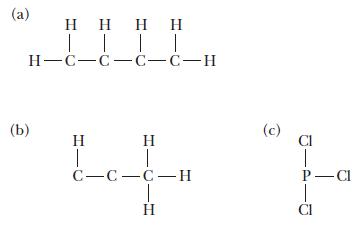
Transcribed Image Text:
(a) (b) |||| H-C-C-C-C-H H 1 H T C-C-C-H (c) Cl T P-Cl Cl
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
H H H H b120 120 120 ...View the full answer

Answered By

Danish Sohail
My objective is to become most reliable expert for clients. For last 10 years I have been associated with the field of accounting and finance. My aim is to strive for best results and pay particular attention to client needs. I am always enthusiastic to help clients for issues and concerns related to business studies. I can work on analysis of the financial statements, calculate different ratios and analysis of ratios. I can critically evaluate stock prices based on the financial analysis and valuation for companies using financial statements of the business entity being valued with use of excel tools. I have expertise to provide effective and reliable help for projects in corporate finance, equity investments, financial accounting, cost accounting, financial planning, business plans, marketing plans, performance measurement, budgeting, economic research, risk assessment, risk management, derivatives, fixed income investments, taxation, auditing, and financial performance analysis.
4.80+
78+ Reviews
112+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
For each of the following molecules, complete the Lewis structure and use the VSEPR model to determine the bond angles around each central atom. Note that the drawings are only skeleton structures...
-
For each of the following molecules, complete the Lewis structure and use the VSEPR model to determine the bond angles around each central atom. Note that the drawings are only skeleton structures...
-
For each of the following molecules, complete the Lewis structure and use the VSEPR model to determine the bond angles around each central atom. Note that the drawings are only skeleton structures...
-
(a) Write an equation describing a sinusoidal transverse wave traveling on a cord in the positive direction of a y axis with an angular wave number of 60 cm-1, a period of 0.20 s, and an amplitude of...
-
Sales and Growth the most recent financial statements for Heng Co. are shown here: Assets and costs are proportional to sales. The company maintains a constant 30 percent dividend payout ratio and a...
-
Yuka consumes mangos and oranges. She is given four mangos and three oranges. She can buy or sell mangos for $ 2 each. Similarly, she can buy or sell an orange for $ 1. If Yuka has no other source of...
-
Could an include use case have a secondary actor? Explain.
-
The time between calls to a corporate office is exponentially distributed with a mean of 10 minutes. (a) What is the probability that there are more than three calls in one-half hour? (b) What is the...
-
Crane Company issued $700000 of 8%, 5-year bonds at 104, Assuming straight-line amortization and annual interest payments, what is the amount of the amortization at each interest payment date?
-
Use the VSEPR model to predict the bond angles around each central atom in the following Lewis structures (benzene rings are frequently pictured as hexagons, without the letter for the carbon atom at...
-
Consider the following molecules: (a) CH 4 (b) NH 2 Cl (c) BF 3 (d) CS 2 (i) Which compound has the most polar bonds? (ii) Which compounds in the list are not polar?
-
Prove that the area of this region in the plane is equal to the value of this determinant. Compare with this. 42 1 det(X2 y1 det/ ( 2
-
Pick your favorite brand and conduct a digital media audit for that brand. This involves monitoring which networks the brand is active on, the types of content posted on each network, and the level...
-
Based on the content posted by Burton on Instagram and Facebook what contents are posted by consumers ? What are the key issues found ? What do you suggest the brand do to connect better with its...
-
Future Value of Multiple Annuities Assume that you contribute $220 per month to a retirement plan for 20 years. Then you are able to increase the contribution to $320 per month for another 20 years....
-
1. The course of social media strategy on hubspot.com identifies four general categories of social media KPIs, namely Reach Engagement Return on investment (ROI) Retention and loyalty SELECT TWO...
-
You are asked to come up with a piecewise function f(x) that includes a maximum of four functions that satisfy the following conditions and then provide a Desmo graph of this piecewise function. The...
-
Where would you expect to find a larger percentage of childless couples: at a cheap movie or at an expensive show?
-
The following items were displayed in the statement of affairs for Lubbock Company: Fully secured liabilities ......... $90,000 Partially secured liabilities ....... 12,000 Unsecured liabilities...
-
The shaft has a radius c and is subjected to a torque per unit length of t 0 , which is distributed uniformly over the shafts entire length L. If it is fixed at its far end A, determine the angle of...
-
The 60-mm-diameter solid shaft is made of A-36 steel and is subjected to the distributed and concentrated torsional loadings shown. Determine the angle of twist at the free end A of the shaft due to...
-
The contour of the surface of the shaft is defined by the equation y = e ax , where a is a constant. If the shaft is subjected to a torque T at its ends, determine the angle of twist of end A with...
-
Cobe Company has manufactured 295 partially finished cabinets at a cost of $73,750. These can be sold as is for $88,500. Instead, the cabinets can be stained and fitted with hardware to make finished...
-
Consider the above graph: find a minimum spanning tree MST of the graph by running Kurskal's algorithm. a) What is the total weight of the MST? b) List the edges of the MST
-
Rodriguez Company pays $315,900 for real estate with land, land improvements, and a building. Land is appraised at $200,000; land improvements are appraised at $50,000; and the building is appraised...

Study smarter with the SolutionInn App


