For each of the following molecules, complete the Lewis structure and use the VSEPR model to determine
Question:
For each of the following molecules, complete the Lewis structure and use the VSEPR model to determine the bond angles around each central atom. Note that the drawings are only skeleton structures and may depict the angles incorrectly.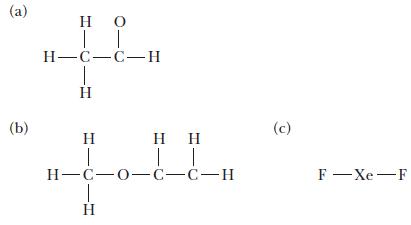
Transcribed Image Text:
(a) (b) IT H=C=C= H IT H-C-0-C-C-H (c) F - Xe-F
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
a H O I HCCH I H 1095 arou...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
For each of the following molecules, complete the Lewis structure and use the VSEPR model to determine the bond angles around each central atom. Note that the drawings are only skeleton structures...
-
For each of the following molecules, complete the Lewis structure and use the VSEPR model to determine the bond angles around each central atom. Note that the drawings are only skeleton structures...
-
For each of the following molecules, complete the Lewis structure and use the VSEPR model to determine the bond angles around each central atom. Note that the drawings are only skeleton structures...
-
Ratio Computation and Analysis; Liquidity) as loan analyst for Madison Bank, you have been presented the following information. Each of these companies has requested a loan of $50,000 for 6 months...
-
Industry-Specific Ratios Specialized ratios are sometimes used in specific industries, For example, the so-called book-to-bill ratio is closely watched for semiconductor manufacturers. A ratio of .93...
-
Legendary for his business and investment acumen, Warren Buffett is frequently called the Oracle of Omaha after his birthplace in Omaha, Nebraska. As one of the worlds wealthiest business...
-
What are the four concepts that are essential to the extended object-oriented approach to software development?
-
Data pertaining to the current position of Boole Company are as follows: Cash ................ $240,000 Temporary investments ......... 120,000 Accounts and notes receivable (net) ... 360,000...
-
(QUESTIONS) 1. Facts of the Case Provide a very concise and clear summary of the case. Be brief but do include all information that you feel is essential to the case. 2. Arguments provided by the...
-
Indicate which of the following molecules are polar. Draw the molecular structure of each polar molecule, including the arrows that indicate the bond dipoles and the molecular dipole moment. (a) NF 3...
-
The molecular orbital diagram of NO shown in Figure 10.47 also applies to OF - . Draw the complete molecular orbital diagram for OF - . What is the OF bond order? Figure 10.47 Energy Atomic orbitals...
-
The Doral Company manufactures and sells pens. Present sales output is 5,000,000 units per year at a selling price of $0.60 per unit. Fixed costs are $1,080,000 per year. Variable costs are $0.36 per...
-
Let's assume that someone does triple encryption by using EEE with CBC on the inside. Suppose an attacker modifies bit x of ciphertext block n. How does this affect the decrypted plaintext?
-
Before you can begin to design a network, you must take into account the needs of the organization and the people associated with it as well as how those people will be accessing the network. For...
-
Do you see someone skilled in their work? They will stand before kings; They will not serve before officials of low rank" (NIV). This verse can be used as an encouragement to do everything that one...
-
As a Network Administrator, you will be responsible for many IT functions of your organization including, but not limited to, Active Directory installation, Active Directory object...
-
Receiving feedback can be challenging but doing so can be the perfect practice ground for giving it. This week,youhavelearned how critical feedback can be in the workplace, affecting everything from...
-
What is float?
-
Portal Manufacturing has total fixed costs of $520,000. A unit of product sells for $15 and variable costs per unit are $11. a). Prepare a contribution margin income statement showing predicted net...
-
The shaft is supported by smooth journal bearings at A and B that only exert vertical reactions on the shaft. Determine its smallest diameter d if the allowable bending stress is Ï allow = 180...
-
The axle of the freight car is subjected to a wheel loading of 20 kip. If it is supported by two journal bearings at C and D, determine the maximum bending stress developed at the center of the axle,...
-
The strut on the utility pole supports the cable having a weight of 600 lb. Determine the absolute maximum bending stress in the strut if A, B, and C are assumed to be pinned. 2 in. -4 ft- -2 ft C01...
-
Whispering Winds Company exchanges old delivery equipment for new delivery equipment. The book value of the old delivery equipment is $31,900 (cost $61,900 less accumulated depreciation $30,000). Its...
-
The Wildhorse Corporation has 142,000 $2.00 noncumulative preferred shares that have been issued. It declares a quarterly cash dividend on May 15 to shareholders of record on June 10. The dividend is...
-
Find (f+g)(-2) when f(x)=x+2 and g(x) = x - 1.

Study smarter with the SolutionInn App


