The molecular orbital diagram of NO shown in Figure 10.47 also applies to OF - . Draw
Question:
The molecular orbital diagram of NO shown in Figure 10.47 also applies to OF-. Draw the complete molecular orbital diagram for OF-. What is the OF bond order?
Figure 10.47 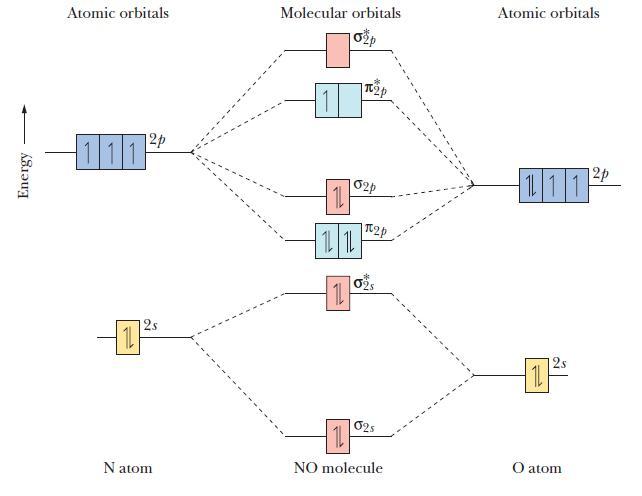
Transcribed Image Text:
Energy Atomic orbitals 12p 111 12s Natom Molecular orbitals 11 1L 1L 11 p 02p 2p 02S 025 11 NO molecule Atomic orbitals 111 11 2 12p O atom
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
While I cannot draw and provide you the complete molecular orbital diagram for OF here I can explain how to construct one based on the provided diagra...View the full answer

Answered By

Emel Khan
I have the ability to effectively communicate and demonstrate concepts to students. Through my practical application of the subject required, I am able to provide real-world examples and clarify complex ideas. This helps students to better understand and retain the information, leading to improved performance and confidence in their abilities. Additionally, my hands-on approach allows for interactive lessons and personalized instruction, catering to the individual needs and learning styles of each student.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
The molecular orbital diagram of NO shown in Figure 10.47 also applies to CO. Draw the complete molecular orbital diagram for CO. What is the CO bond order? Figure 10.47 Energy Atomic orbitals 12p...
-
The molecular orbital diagram of NO shown in Figure 10.47 also applies to the following species. Write the molecular orbital electron confi guration of each, indicating the bond order and the number...
-
The nitrosyl ion, NO + , has an interesting chemistry. Assume the molecular orbital diagram shown in Figure 9.16 applies to NO + . (a) Is NO + diamagnetic or paramagnetic? If paramagnetic, how many...
-
Northland Corporation is a small information-systems consulting firm that specializes in helping companies implement standard sales-management software. The market for Northalndss services is very...
-
Du Pont Identity Why is the Du Pont identity a valuable tool for analyzing the performance of a firm discuss the types of information ii reveals compared to ROE considered by itself.
-
Two firms, A and B, produce goods A and B, respectively. The linear demands for the two goods are, respectively, QA = 100 4PA + 1.5PB QB = 120 2PB + 0.5PA Production costs are constant but not...
-
What are the factors that shape software modeling?
-
Intermountain Resources is a multidivisional company. It has three divisions with the following betas and proportion of the firms total assets: The risk-free rate is 7 percent and the market risk...
-
Any areas of opportunity in terms of product categories, marketing, or target audiences that would expand that brand "Vision Streetwear". what are 3 key strengths of the brand to highlight and expand...
-
For each of the following molecules, complete the Lewis structure and use the VSEPR model to determine the bond angles around each central atom. Note that the drawings are only skeleton structures...
-
The delocalized bonding that describes O 3 also applies to NO 2 . Draw the delocalized molecular orbital for NO 2 .
-
Nitrites (RC ? N) react with Grignard reagents (R'MgBr). The reaction product from 2-methyipropanenitrile with methyl magnesium bromide has the following spectroscopic properties. Propose a...
-
1. Identify a team that you were (or are) a member of. Describe the role the leader played in the team's success (or failure). 2. Explain how the leader dealt with any conflicts or disagreements that...
-
Apply these questions to The Home Depot. What segment of the macro environment has the strongest impact (beneficial or detrimental) on this company's industry? Are all companies in the industry...
-
Two (2) high ranking managers of Anrun Corp. know that the company's revenue is rapidly declining. However, at a recent shareholder meeting, they tell the shareholders to expect record profits in the...
-
Kathy is the project manager, and the project concerns with developing an active prosthetic arm. Kathy is responsible for writing the human resources management plan. Part of the plan consist s of...
-
Sunrise Medical Center is the largest healthcare facility in the southern region of the United States. With a 2,500-bed count, the medical center sees 800,000 - 900,000 patients in a year. Their...
-
What does the WACC for a firm tell us?
-
The Higher the time period of the financial security the higher the. ............... risk. O a. Maturity O b. Default and Maturity Oc. Default O d. Liquidity
-
The boat has a weight of 2300 lb and a center of gravity at G. If it rests on the trailer at the smooth contact A and can be considered pinned at B, determine the absolute maximum bending stress...
-
Determine the absolute maximum bending stress in the 1.5-in.-diameter shaft. The shaft is supported by a thrust bearing at A and a journal bearing at B. 400 Ib 300 lb 12 in 18 in. 15 in.
-
Determine the smallest allowable diameter of the shaft. The shaft is supported by a thrust bearing at A and a journal bearing at B. The allowable bending stress is Ï allow = 22 ksi. 400 lb 300...
-
Following is information on two alternative investment projects being considered by Tiger Company. The company requires an 8% return from its investments. (PV of $1, FV of $1, PVA of $1, and FVA of...
-
The St. Louis to Seattle Railroad is considering acquiring equipment at a cost of $212,000. The equipment has an estimated life of 10 years and no residual value. It is expected to provide yearly net...
-
The Platter Valley factory of Bybee Industries manufactures field boots. The cost of each boot includes direct materials, direct labor, and manufacturing (factory) overhead. The firm traces all...

Study smarter with the SolutionInn App


