For each of the reactions, calculate E from the table of standard potentials, and state whether the
Question:
For each of the reactions, calculate E° from the table of standard potentials, and state whether the reaction is spontaneous as written or spontaneous in the reverse direction under standard conditions.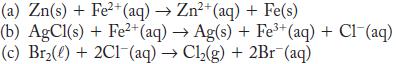
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
a E 136 V spontaneous in the ...View the full answer

Answered By

Muhammad Haroon
More than 3 years experience in teaching undergraduate and graduate level courses which includes Object Oriented Programming, Data Structures, Algorithms, Database Systems, Theory of Automata, Theory of Computation, Database Administration, Web Technologies etc.
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
For each of the reactions, calculate E from the table of standard potentials, and state whether the reaction is spontaneous as written or spontaneous in the reverse direction under standard...
-
Calculate the potential for each of the voltaic cells in Exercise 18.44 when the concentrations of the soluble species and gas pressures are as follows: Exercise 18.44 For each of the reactions,...
-
(a) LEP Table 12-2: Exothermic Reaction with Heat Exchange Download the Polymath, MATLAB, Python, or Wolfram codes for the algorithm and data given in Table T12-2 for the exothermic gas phase...
-
Which of the following options are available for creating a policy in Qualys Policy Compliance? (Choose three) A, Create from Host B, Create from Scratch C, Import from Library D, Import from CSV File
-
Growers use giant fans to prevent grapes from freezing when the effective sky temperature is low. The grape, which may be viewed as a thin skin of negligible thermal resistance enclosing a volume of...
-
For hierarchical routing with 4800 routers, what region and cluster sizes should be chosen to minimize the size of the routing table for a three-layer hierarchy? A good starting place is the...
-
What is the typical number of harmonics of running speed in a spectrum taken on a machine operating within satisfactory limits?
-
After-Tax Cost of Debt LL Incorporateds currently outstanding 11% coupon bonds have a yield to maturity of 8%. LL believes it could issue at per new bonds that would provide a similar yield to...
-
Carlberg Company has two manufacturing departments, Assembly and Painting. The Assembly department started 10,700 units during November. The following production activity in both units and costs...
-
Use the data from the table of standard reduction potentials in Appendix H to calculate the standard potential of the cell based on each of the following reactions. In each case, state whether the...
-
Two electrodes are immersed in a 1 M HBr solution. One of the electrodes is a silver wire coated with a deposit of AgBr(s). Th e second electrode is a platinum wire in electrical contact with a...
-
Healthcheck Corp. manufactures an antacid product that passes through two departments. Data for June for the first department follow: The beginning work in process inventory was 80% complete with...
-
Give example goals for an online acquisition campaign in terms of response rates or engagement with creative, cost and overall campaign effectiveness.
-
How are customers expectations formed? Explain the difference between desired service and adequate service with reference to a service experience youve had recently.
-
Explain the term prototyping in relation to website creation.
-
Briefly summarise the implications of the Internet on each of these elements of the marketing mix: a Product b Price c Place d Promotion.
-
What are the main reasons for the growing share of the service sector in all major economies of the world?
-
(Multiple Choice) 1. The Unearned Revenue account of Lorelai Incorporated began 2012 with a normal balance of $5,000 and ended 2012 with a normal balance of $15,000. During 2012, the Unearned Revenue...
-
Coastal Refining Company operates a refinery with a distillation capacity of 12,000 barrels per day. As a new member of Coastal's management team, you have been given the task of developing a...
-
(a) Suppose a tire rolls without slipping on a horizontal road. Explain the role friction plays in this motion. What two surfaces are involved in this frictional force? Is it static friction or...
-
You are a newly graduated astronaut preparing for your first trip into space. Plans call for your spacecraft to reach a velocity of 500 m/s after 2.4 min. If your mass is 75 kg, what force will be...
-
The lower piece of silk in Figure 3.20 is acted on by two forces, +T 2 at the upper end and -T 2 at the lower end. These two forces are equal in magnitude and opposite in direction. Are they an...
-
You decide to arrange a meeting so that internal and external stakeholders can have input into the development of a service program for at-risk youth in the community. One of the stakeholders you...
-
In Java script needed Use indexOf(), and substr() or substring(), to display the end of the sentence starting from the word "Rome". let wiseProverb = "When in Rome do as Romans do."; // Code will be...
-
You are to conduct an interview with General Eugene Irwin. You are to assume the role of his character and answer the following three questions. Movie: The Last Castle 2001 1. Why do you believe you...

Study smarter with the SolutionInn App


