Given the following two equations, where the metals (M and M') are from the second period, what
Question:
Given the following two equations, where the metals (M and M') are from the second period, what are the metals, and which metal cation in the product has a larger radius?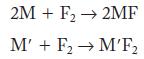
Transcribed Image Text:
2M + F₂ → 2MF M' + F₂ → M'F₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
2LiF Be F 2 LiF 5 Lithium fluorid...View the full answer

Answered By

GERALD KAMAU
non-plagiarism work, timely work and A++ work
4.40+
6+ Reviews
11+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Write a literature review for your study. See below for an example of a literature review. Your literature review should provide both analysis and synthesis of previous studies as related to the...
-
1. In the Amazon.com example of free shipping, the initial experience was different in France because in that country_________, 2. The difference between the Pepsi and Coke taste challenge campaigns...
-
Last year quite a few fast food restaurants (Wendys, McDonalds, Churches, Popeyes and others) engaged in a competitive battle for customers with chicken sandwiches . What marketing strategies did...
-
The 2019 catalog that Gigantic State University (GSU) sent to prospective students described a merit-based scholarship called the Eagle Scholarship. The catalog stated that GSU offers the Eagle...
-
Gallium arsenide (GaAs) and gallium phosphide (GaP) are compound semiconductors that have room-temperature band gap energies of 1.42 and 2.25 eV, respectively, and form solid solutions in all...
-
Figure 2-36 is a position versus time graph for the motion of an object along the x axis. Consider the time interval from A to B (a) Is the object moving in the position or negative direction? (b) Is...
-
Project Scope Management Create a WBS for this project and enter the tasks in Project 2010. Create milestones and summary tasks. Assume that the main WBS categories and some of the project management...
-
Williams Glassware has estimated, at various debt ratios, the expected earnings per share and the standard deviation of the earnings per share as shown in the following table. a. Estimate the optimal...
-
What is the first step you will take to successfully complete a budget for yourself? What are some of the challenges in creating a budget for a company?
-
Sunnyville Bank wants to identify customers who may be interested in its new mobile banking app. The worksheet called Mobile_Banking_Data contains 500 customer records collected from a previous...
-
Write the electron confi guration of the alkali metal (M) that reacts with oxygen to yield an oxide, M2O, if 1.22 g of the metal reacts with 1.41 g of oxygen to form 2.63 g of the oxide.
-
What masses of iron and Cl 2 are needed to prepare 7.88 g of the metal halide product if, under the conditions of the reaction, the electron confi guration of the iron cation in the product is...
-
From a purely financial perspective, are there situations in which a business would be better off choosing a project with a shorter payback over one that has a larger NPV?
-
In Exercises 29 and 30, find the probabilities and indicate when the "5% guideline for cumbersome calculations" is used. 29. Medical Helicopters In a study of helicopter usage and patient survival,...
-
Introduction to Internetworking Project 1: Ctrl-Alt-Del Inc. INTRODUCTION You have accepted a contract to participate in the design of the network infrastructure of a company called Ctrl-Alt-Del Inc....
-
Construct Arguments Tell whether each statement is always true, sometimes true, or never true. Explain. a. An integer is a whole number. b. A natural number is a rational number. c. An irrational...
-
Please answer the following Questions : 1. Who are the competitors for Whole Foods? 2. Do you consider traditional supermarkets to be competitors for natural and organic supermarkets? 3. How would...
-
LNC Corp is trying to determine the effect of its inventory turnover ratio and DSO on its cash conversion. Credit sales in 2016 is $101,000, cost of goods sold will be 70% of sales and it earned a...
-
A transient numerical analysis is to be performed on the composite material section shown in Figure P4-86. Calculate the maximum time increment that can be used for node 5 to ensure convergence....
-
When you weigh yourself on good old terra firma (solid ground), your weight is 142 lb. In an elevator your apparent weight is 121 lb. What are the direction and magnitude of the elevator's...
-
Draw a Haworth projection for each of the following compounds: (a) -d-Galactopyranose (b) -d-Mannopyranose (c) -d-Allopyranose (d) -d-Mannopyranose (e) -d-Glucopyranose (f ) -d-Glucopyranose
-
Provide a complete name for the following compound: CH-
-
Mutarotation causes the conversion of b-d-mannopyranose to -d-mannopyranose. Using Haworth projections, draw the equilibrium between the two pyranose forms and the open-chain form of d-mannose.
-
Describe how the following affect the valuation of PPE. a) Cash Discounts b) Deferred Payment Contracts
-
Lou Barlow, a divisional manager for Sage Company, has an opportunity to manufacture and sell one of two new products for a five - year period. His annual pay raises are determined by his division s...
-
Consider a 5 year debt with a 15% coupon rate paid semi-annually, redeemable at Php1,000 par. The bond is selling at 90%. The flotation cost is Php50 per bind. The firm's tax bracket is 30%.

Study smarter with the SolutionInn App


