Identify the orbitals that overlap to form the bonds in HCN. HIS- C 5/3 C C
Question:
Identify the orbitals that overlap to form the bonds in HCN.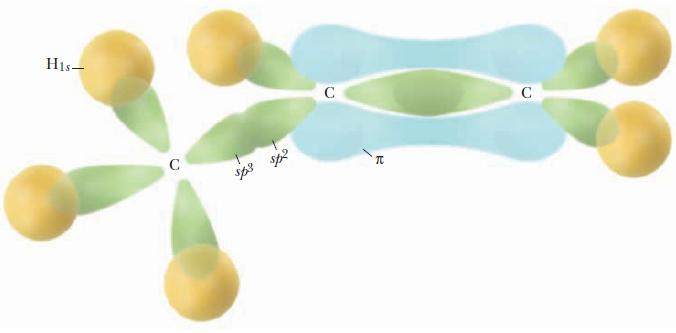
Transcribed Image Text:
HIS- C 5/3 C C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 25% (4 reviews)
The sphybridized carbon atom forms two bonds one by overl...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Draw the two resonance structures that describe the bonding in the acetate ion. What is the hybridization of the carbon atom of the CO 2 group? Select one of the two resonance structures and...
-
Classify each of the labeled bonds in the following structure in terms of the bond type ( or ) and the component orbitals that overlap to form the bond. (For example, the carbon--carbon bond in...
-
Classify each of the labeled bonds in the following structure in terms of the bond type ( or ) and the component orbitals that overlap to form the bond. (For example, the carboncarbon bond in ethane...
-
In the figure below, a square of edge lengths is formed by four spheres of masses, m, M, m3, and m4. What is the x component and the y component of the net gravitational force from them on a central...
-
The harmonic decomposition problem considered by Pisarenko can be expressed as the solution to the equation. The solution for a can be obtained by minimizing the quadratic form a*l?yya subject to the...
-
Altoona Valve Companys planned production for the year just ended was 20,000 units. This production level was achieved, and 21,000 units were sold. Other data follow: Direct material...
-
Develop the questionnaire in Internet Exercise 8-21 using an electronic questionnaire design package that is available for free (see the websites mentioned in this chapter in the subsection entitled...
-
Ehlo Company is a multiproduct firm. Presented below is information concerning one of its products, the Hawkeye. Instructions Compute cost of goods sold, assuming Ehlo uses: (a) Periodic system, FIFO...
-
difference between index and index fund difference between uits and eitfs give an example from pakistani market
-
Identify the hybridization of the central atom and the orbitals used for each bond in IF 5 . Strategy The steric number, determined from the Lewis structure, defines the hybrid orbitals needed for a...
-
Describe the bonds in propylene, CH 3 CH=CH 2 . Identify the hybridization of each central atom and the type ( or ) of each bond. Strategy Use the steric numbers determined from the Lewis structure...
-
The variable expressions represent the angle measures of a triangle. Find the measure of each angle. Then classify the triangle by its angles. MLA = x mLB = (4x) mLC = (5x)
-
Does a cation gain protons to form a positive charge or does it lose electrons? a) The protons in the nucleus of a cation formed due to the loss of protons and electrons. b) The cation gains protons...
-
Select the coefficients necessary to balance each equation. Choose a coefficient for every compound. 1. Choose... NH4NO3 2. Choose... Choose... Fe + Choose... H Choose... NO + Choose... HO FeCl3 +...
-
Identify the number of structural isomers that have the formula, C3H8. A) 1 B) 2 C) 3 D) 4 E) 5
-
1. It is possible on the basis of shell model that nucleus with Z = 110 and A = 294 may be exceptionally long-lived. Estimate its nuclear radius.
-
9. Compute the electrical change on a bit x (x,y) x (x, y) q (0,0) -8 (0,)
-
Why would the transactions motive and the precautionary motive for holding money both tend to vary directly with the price level? Why would the quantity of money people desire to hold for both...
-
Catherine (aged 42) and Johnson (aged 45) have been married for 12 years. Johnson is a project manager of an event company at a monthly salary of $55,000 with an additional one-month salary of...
-
Determine the shear strain γ xy at corners A and B if the plate distorts as shown by the dashed lines. 5 mm, 2 mm 4 mm IB 2 mm 300 mm T2 mm -X- 400 mm 3 mm
-
Determine the shear strain γ xy at corners D and C if the plate distorts as shown by the dashed lines. 5 mm, 2 mm 4 mm IB 2 mm 300 mm T2 mm 400 mm- 3 mm
-
The pipe with two rigid caps attached to its ends is subjected to an axial force P. If the pipe is made from a material having a modulus of elasticity E and Poissons ratio v, determine the change in...
-
I have a measurement of variable that is normally distributed with a mean of 2 0 and a standard deviation of 5 . What is the score associated with the 6 5 th percentile in the population if we appeal...
-
A windmill has blades that are 14 feet long. If the windmill is rotating at 5 revolutions per second, find the linear speed of the tips of the blades in miles per hour.
-
A merry go round rotates 2808 degrees per ride. How far would a rider seated 8 feet from the center of the merry-go round travel during the ride

Study smarter with the SolutionInn App


