In the beakers shown below, the colored spheres represent a particular ion, with the dark gray balls
Question:
In the beakers shown below, the colored spheres represent a particular ion, with the dark gray balls representing Pb2+. In one reactant beaker is Pb(NO3)2 and in the other is NaCl. In the product beaker, the organized solid represents an insoluble compound. Write the overall equation, the complete ionic equation, and the net ionic equation.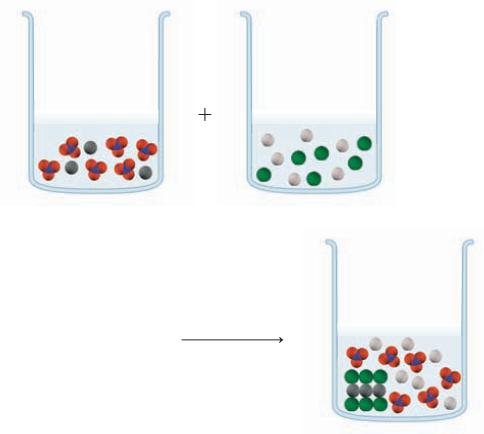
Transcribed Image Text:
+
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
overall Pb NO32aq 2NaClaq ...View the full answer

Answered By

Antony Mutonga
I am a professional educator and writer with exceptional skills in assisting bloggers and other specializations that necessitate a fantastic writer. One of the most significant parts of being the best is that I have provided excellent service to a large number of clients. With my exceptional abilities, I have amassed a large number of references, allowing me to continue working as a respected and admired writer. As a skilled content writer, I am also a reputable IT writer with the necessary talents to turn papers into exceptional results.
4.50+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
In the beakers shown below, the colored spheres represent a particular ion, with the dark gray balls representing Ag + . In one reactant beaker is AgNO 3 and in the other is NaBr. In the product...
-
Shown below is the cubic unit cell of an ionic compound. Answer the questions by referring to this structure. Be careful to note that some atoms are hidden by those in front. a. One of the spheres...
-
Part 1 a. Ammonia, NH 3 , is a weak electrolyte. It forms ions in solution by reacting with water molecules to form the ammonium ion and hydroxide ion. Write the balanced chemical reaction for this...
-
Amie, Inc., has 100,000 shares of $2 par value stock outstanding. Prairie Corporation acquired 30,000 of Amie's shares on January 1, 2015, for $120,000 when Amie's net assets had a total fair value...
-
Estimate the minimum frequency of a -ray that causes a deuteron to disintegrate into a proton and a neutron, commenting on any assumptions you make. The masses of the particles are md = 2.0141mu, mp...
-
a. Find the inverse of the function (x) = mx, where m is a constant different from zero. b. What can you conclude about the inverse of a function y = (x) whose graph is a line through the origin with...
-
The horizontal portion of the wire in Figure P28.76 has a length \(\ell=0.100 \mathrm{~m}\), and position \(\mathrm{P}\) is a perpendicular distance \(d=30.0 \mathrm{~mm}\) above the center of the...
-
In the normal course of performing their responsibilities, auditors often conduct audits or reviews of the following: 1. Federal income tax returns of an officer of the corporation to determine...
-
24. A water jet whose cross section area is a striker wall making an angle e with the normal and rebounds elastically. The velocity of water of density d is v. Force exerted on wall is :- 0 (1) 2avd...
-
Benito Mendez opened Mendez Appraisals. He rented office space and has a part-time secretary to answer the telephone and make appraisal appointments. His chart of accounts is as follows: Mendezs...
-
An aqueous sample is known to contain either Pb 2+ or Fe 3+ ions. Treatment of the sample with Na 2 SO 4 produces a precipitate. Use the solubility rules to determine which cation is present.
-
An aqueous sample is known to contain either Mg 2+ or Ba 2+ ions. Treatment of the sample with Na 2 CO 3 produces a precipitate, but treatment with ammonium sulfate does not. Use the solubility rules...
-
Plaintiff, a Swiss corporation, entered into contracts to purchase chicken from B.N.S. International Sales Corporation. Defendant was a New York corporation. The English language contracts called for...
-
Define the variance of a discrete random variable, and explain in what sense the values of a random variable are more spread out if the variance is larger.
-
Explain the difference between economic and statistical significance.
-
Explain how the \(p d f\) of a continuous random variable is different from the \(p d f\) of a discrete random variable.
-
Give an intuitive explanation of statistical independence of two random variables, and state the conditions that must hold to prove statistical independence. Give examples of two independent random...
-
In a capital murder trial, with a potential penalty of life in prison, would you as judge tell the jury to make sure that we accidently convict an innocent person only one time in a hundred, or use...
-
Fluent, an investor in stocks and bonds wanted to increase his portfolio but wanted to minimize his tax liability on the income from the bonds. He is presented with the following alternative...
-
Identify the source of funds within Micro Credit? How does this differ from traditional sources of financing? What internal and external governance mechanisms are in place in Micro Credit?
-
Use LHpitals rule lim [f(x)/g(x)] x 0 = lim [df(x)dx/dg(x)/dx] x 0 to show that the expression derived for Pf in part b of Example problem 1.1 hane the correct limit as y 0.
-
For each compound below, identify all lone pairs and indicate whether each lone pair is localized or delocalized. Then, use that information to determine the hybridization state and geometry for each...
-
Nicotine is a toxic substance present in tobacco leaves. There are two lone pairs in the structure of nicotine. In general, localized lone pairs are much more reactive than delocalized lone pairs....
-
Question 16 of 16 Current Attempt in Progress Calculate activity-based costing rates from the following budgeted data for Kimberly's Tools: Activity Cost Pool Budgeted Cost Budgeted Cost Driver...
-
ERD with these questions. What type of account(s) do you have with Noble Bank? a. Checking Account b. Savings Account c. Credit Card Account d. Other How many active accounts and or cards do you...
-
Common stock Additional paid-in capital Total paid-in capital Retained earnings 800 800 4,200 4,200 5,000 5,000 29,880 26,660 Total stockholders' equity 34,880 31,660 Total liabilities and...

Study smarter with the SolutionInn App


