Nicotine is a toxic substance present in tobacco leaves. There are two lone pairs in the structure
Question:
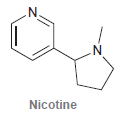
There are two lone pairs in the structure of nicotine. In general, localized lone pairs are much more reactive than delocalized lone pairs. With this information in mind, do you expect both lone pairs in nicotine to be reactive? Justify your answer.
Transcribed Image Text:
Nicotine
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (14 reviews)
Both lone pairs ...View the full answer

Answered By

Gilbert Chesire
I am a diligent writer who understands the writing conventions used in the industry and with the expertise to produce high quality papers at all times. I love to write plagiarism free work with which the grammar flows perfectly. I write both academics and articles with a lot of enthusiasm. I am always determined to put the interests of my customers before mine so as to build a cohesive environment where we can benefit from each other. I value all my clients and I pay them back by delivering the quality of work they yearn to get.
4.80+
14+ Reviews
49+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
White phosphorus is much more reactive than black or red phosphorus. Explain.
-
Why is trityl chloride much more reactive than the other alkyl halides in Table 17.2? TABLE 17 2 comparison of S,1 Solvolysis Rates of Benzylic and Nonbenzylic Alkyl Halides 25C R-CIHo l + H2O -OH +...
-
Ethyl triflate is much more reactive than ethyl mesylate toward nucleophiles in SN2 reactions. (a) Give the structures of all of the products formed when each compound reacts with potassium iodide in...
-
You bought a share of 3.4 percent preferred stock for $96.82 last year. The market price for your stock is now $98.34. What is your total return for last year?
-
This cases focuses on the issue of performance. As discussed in the text, clear and effective goals help improve employee performance, reduce role stress conflict and ambiguity, and improve the...
-
A heavy rock and a light rock in free fall (zero air resistance) have the same acceleration. The reason the heavy rock doesnt have a greater acceleration is that the (a) force due to gravity is the...
-
Petal Length and Petal Width Figure 2.93 (a) shows a scatterplot of the two quantitative variables petal length and petal width. (a) Explain how the scatterplot appears to show at least two different...
-
A companys 5-year bonds are yielding 7.75% per year. Treasury bonds with the same maturity are yielding 5.2% per year, and the real risk-free rate (r*) is 2.3%. The average inflation premium is 2.5%;...
-
Leveraged Investing with Eligible Dividends $50,000 Cash purchase + $50,000 borrowed funds at 4.95% 12 month holding period Eligible monthly dividend of $0.07/share Purchase price of $25.00 Sale...
-
1. How does Amazon.com illustrate the sources of service sector growth? Comment on information technology, Internet as an enabler, innovation, and changing demographics. 2. What generic approaches(s)...
-
For each compound below, identify all lone pairs and indicate whether each lone pair is localized or delocalized. Then, use that information to determine the hybridization state and geometry for each...
-
A 455 cm 3 vessel contains a mixture of Ar and Xe. If the mass of the gas mixture is 2.245 g at 25.0C and the pressure is 760. Torr, calculate the mole fraction of Xe in the mixture.
-
Yolanda and Xavier, spouses, have four adult children, Andy, Betty, Cathy, and Danny. In 2014, they made a number of gifts. Yolanda gave Andy cash of $40,000 and Betty stock valued at $60,000. Xavier...
-
Crane Corporation began operations in 2025. During the year, the company earned sales revenues of $1686800 with related cost of goods sold of $858600. The company incurred administrative expenses of...
-
Vaughn Corporation made credit sales of $39,600 which are subject to 5% sales tax. The corporation also made cash sales which totaled $16,905 including the 5% sales tax. (a) Prepare the entry to...
-
An income producing asset costs $50,706 and will be depreciated using Double Declining Balance with a $3,363 salvage value over 9 years. What is the Book Value in year 2?
-
1 Find the compound amount and compound interest if $2000 is invested for six years and interest is compounded continuously at the annual rate 7% The compound amount will be $ 3136.62 (Round to the...
-
Your company receives 40 excess copies of a book. You plan to sell these books in two markets: directly in the store or sell them on eBay. Here are the two demand equations: Store Demand: Ps = 200 -...
-
Find an equation for graph. 2 m/2
-
Under what conditions is the following SQL statement valid?
-
Make a reaction flowchart (roadmap diagram), as in previous problems, to organize the information provided to solve this problem. An optically active compound A (assume that it is dextrorotatory) has...
-
A naturally occurring antibiotic called mycomycin has the structure shown here. Mycomycin is optically active. Explain this by writing structures for the enantiomeric forms of mycomycin....
-
An optically active compound D has the molecular formula C6H10 and shows a peak at about 3300 cm-1 in its IR spectrum. On catalytic hydrogenation D yields E (C6H14). Compound E is optically inactive...
-
Use factoring by grouping to solve the following equation. 3-522-16r+80=0 Enter the solutions, separated by commas if necessary. Do not use a plus-minus () sign when entering separate answers....
-
What are some intervention policies that make provisions for destitute college or university students in Ethiopia? Please provide Examples
-
Develop a simple application of two- variable linear programming problem to find an optimal product mix in a 'realistic' context. Specify the resources to be considered requirements, and the market...

Study smarter with the SolutionInn App


