The value of K c for the reaction N(g) + 3H(g) 2NH3(g) is 2.00 at 400 C.
Question:
The value of Kc for the reaction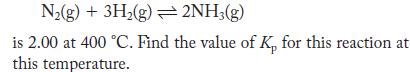
Transcribed Image Text:
N(g) + 3H(g) 2NH3(g) is 2.00 at 400 C. Find the value of K, for this reaction at this temperature.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
Solution N2g 3H2g Kp Kc RT ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
The value of Kc for the following reaction at 900oC is 0.28. What is Kp at this temperature? CS2(g) +4H2(g)CH4(g) + 2H2S(g)
-
Obtain the value of Kc for the following reaction at 900 K: Use the data given in Problem 14.33. 2502(g) + O2(g) 2SO3(g)
-
Obtain the value of Kc for the following reaction at 500 K: Use the data given in Problem 14.34. CO(g) +2H2(g) CH,OH(g)
-
Marquis Company uses a weighted-average perpetual inventorysystem. August 2, 22 units were purchased at $3 per unit. August 18, 27 units were purchased at $5 per unit. August 29, 24 units were sold....
-
Bill Watts, president of Western Publications, accepts a capital budgeting project proposed by Division X. this is the division in which the president spent his first 10 years with the company. ON...
-
Explain how the valuation model given in Equation 1.2 could be used to describe the integrated nature of managerial decision making across the functional areas of business.
-
Explain the statement that applications are solutions to information needs. Provide examples to make your point.
-
Asset Disposal Refer to Exercise 8-6. Assume that Gonzalez Company sold the asset on July 1, 2010, and received $15,000 cash and a note for an additional $15,000. Required 1. Identify and analyze the...
-
Travel agency owners have key salespeople contacts for their suppliers, such as cruise line and tour operator Business Development Managers. Therefore as a professional buyer of travel products, the...
-
In an experiment, 4.95 mol CO 2 , 0.050 mol CO, and 0.050 mol O 2 are placed in a 5.0-L reaction vessel at 1400 K. Calculate the reaction quotient, Q, for the following reaction: If K c for this...
-
Sulfur dioxide reacts with chlorine when sealed in a reactor at increased temperature. At 227 C, K c = 20.9. Calculate K p at this same temperature. SO(g) + Cl(g) SOCl(g)
-
A laminar boundary layer growing along a flat plate is sketched in Fig. P1075C. Several velocity profiles and the boundary layer thickness (x) are also shown. Sketch several streamlines in this flow...
-
The CIB activity of the top five Italian banking groups: similarities and strategic differences Write a paper focusing in particular on Banco BPM, following the following points: 1. The report must...
-
Inflation refers to the increase in general price levels of goods and services over a period. It reduces the purchasing power of money, which can have significant impacts on businesses. For example,...
-
A highly competent MBA 2019 graduate while participating in the CELEBRITY APPRENTICE was given the task of selling a product in the vicinity of Trump Tower in Las Vegas. The same product was given to...
-
What are the demand factors leading to higher ticket prices in the airline industry? Explain how they change the prices. What are the supply factors leading to higher ticket prices in the airline...
-
Roughly 18bn is forecast to be spent in the UK on advertising in 2017. Much of this will be spent by monopolistically competitive firms or oligopolies seeking to differentiate the products they sell...
-
Michael Weber, CFA, is analyzing several aspects of option valuation, including the determinants of the value of an option, the characteristics of various models used to value options, and the...
-
Integration is a vital concept when applied in one?s life. Integrating your life means making ideal choices. Perfect choices on the other go in line with quality decisions. Quality decisions lead to...
-
A car travels along a straight, level road. The car begins a distance x - 25 m from the origin at t - 0.0 s. At t - 5.0 s, the car is at x - 100 m; at t - 8.0 s, it is at x - 300 m. Find the average...
-
Draw a graph showing the position x as a function of time for an object whose acceleration is (a) Constant and positive, (b) Constant and negative, and (c) Positive and increasing with time. Assume...
-
Draw a possible positiontime graph for an object whose velocity as a function of time is described by (a) Figure P2.28 and (b) Figure P2.29. Figure P2.28 Figure P2.29 v (m/s) HHt (s) 200-40060- 0-...
-
Given two sets of m vectors, x; R" and y; ER" for all i = 1,2,,m, verify that the summations 1xx and xy can be vectorized as the following matrix multiplications: where X = [x1 x2 Your answers: m m...
-
Prove if G is a graph and every vertex has degree at least three (i.e., 8(G) 3), then G contains a cycle of even length (i.e., a cycle with an even number of edges).
-
Solve: log(6x-8) = log(2x-4).

Study smarter with the SolutionInn App


