Two 1-L containers at 27 C are connected by a stopcock as pictured below. If each dot
Question:
Two 1-L containers at 27 °C are connected by a stopcock as pictured below. If each dot in the containers represents 0.0020 mol of a nonreactive gas, what is the pressure in each container before and after the stopcock is opened ?Draw the container after the stopcock is opened, indicating the number of red and blue dots on each side.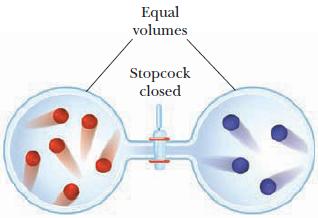
Transcribed Image Text:
Equal volumes Stopcock closed
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
To solve this problem we need to apply the ideal gas law which states PVnRT Where P is the pressure ...View the full answer

Answered By

KRISHNANUNNI K T
I have taught civil engineering subjects like solid mechanics, fluid mechancs, structural analysis etc. individually in coaching centers and professionally for 1 year in a recognized private engineering college. I have done my internship in a structural and geotechnical enginnering firm based in Cochin for about 6 months. I also, worked as a Junior Research Fellow for 3 years in the top ranked NIT of India. I am very much interested in the area of mathematics and have conducted home tutions for ninenth and tenth standard students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Two 1-L containers at 27 C are connected by a stopcock as pictured below. If each dot in the containers represents 0.0050 mol of a nonreactive gas, what is the pressure in each container before and...
-
Containers A and B in FIGURE CP18.73 hold the same gas. The volume of B is four times the volume of A. The two containers are connected by a thin tube (negligible volume) and a valve that is closed....
-
Evaluate lim x 1 + ( 4 x 2 - 1 - x x - 1 ) .
-
Gandhi Ltd renders a promotional service to small retailing businesses. There are three levels of service: the basic, the standard and the comprehensive. On the basis of past experience, the business...
-
If 500 g of molten lead at 327oC is poured into a cavity in a large block of ice at 0oC, how much of the ice melts?
-
What are the advantages and disadvantages of observation studies relative to surveys?
-
The price of a share is \(\$ 40\). If \(\mu=0.1\) and \(\sigma^{2}=0.16\) per year, find a \(95 \%\) confidence interval for the price of the share after six months (i.e. an interval...
-
Account balances taken from the ledger of High Flying Logistics Co. on December 31, 2011, follow: Cash . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . ....
-
In the thin lens approximation, a few simple rules can be used to determine the trajecto- ry of rays after the lens and thus the image formed by the lens: A ray parallel to the optical axis before...
-
Lee Nicholas has been the owner and has operated World.com Advertising since its beginning 10 years ago. The company has prospered. Recently, Nicholas mentioned that he would sell the business for...
-
A robotic analysis of the atmosphere of the planet Venus shows that it has 0.0020. If the total atmospheric pressure on Venus is 91.2 atm, what are the partial pressures (in atm) of each gas? XCO...
-
Sodium metal reacts with water to produce hydrogen gas and sodium hydroxide. Calculate the mass of sodium used in a reaction if 499 mL of wet hydrogen gas are collected over water at 22 C and the...
-
A monitoring system records the production of an assembly line each hour. The design calls for output to be four units per hour. Current policy calls for stopping the line to check for flaws if fewer...
-
Reconsider the situation described in Problem 4. Assume that rather than annual deposits, she makes monthly deposits. The first deposit will be 1 month from today, and the last deposit will be 40...
-
The divestiture technique that Lee is recommending is most likely: A. a spin-off. B. a split-off. C. an equity carve-out. Mark Zin and Stella Lee are CEO and CFO, respectively, of Moonbase...
-
Show that the parity operator \(\Pi\) is its own inverse and is hermitian, so it is unitary.
-
Match the phrases in Column A with the corresponding dividend theory in Column B. Note that you may use the answers in Column B more than once. Column A 1. Bird in the hand 2. Homemade dividends 3....
-
Which policy of the Statement of Corporate Governance is least likely to ensure effective contributions from the board of directors? A. Policy #1. B. Policy #2. C. Policy #3. Mark Zin and Stella Lee...
-
Suppose you are the attorney general of your state. You want to file an action against a gaming corporation You want to seek an injunction against the company, asking it to stop making Internet...
-
An environmentalist wants to determine if the median amount of potassium (mg/L) in rainwater in Lincoln County, Nebraska, is different from that in the rainwater in Clarendon County, South Carolina....
-
Why does it follow from the Heisenberg uncertainty principle that it is not possible to make exact copies of quantum mechanical objects?
-
Which result of the SternGerlach experiment leads to the conclusion that the operators for the z and x components of the magnetic moment do not commute?
-
The Heisenberg uncertainty principle says that the momentum and position of a particle cannot be known simultaneously and exactly. Can that information be obtained by measuring the momentum and...
-
How would you implement your public health campaign on teenage vaping . you may explain two potential barriers that might impact the implementation process and one way you might address one barrier....
-
Please help me with this: In the given reference, which IT function should be segregated from the other function (at least 3)? Why is it important to segregate duties of the following IT functions?...
-
Klijo Limited manufactures three products (A, B and C), with relevant information about each product shown in the table below: Product A B C Demand (units) 220 120 50 Sales price per unit ($) 1440...

Study smarter with the SolutionInn App


