Two 1-L containers at 27 C are connected by a stopcock as pictured below. If each dot
Question:
Two 1-L containers at 27 °C are connected by a stopcock as pictured below. If each dot in the containers represents 0.0050 mol of a nonreactive gas, what is the pressure in each container before and after the stopcock is opened?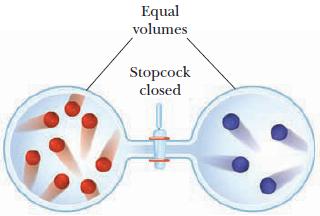
Transcribed Image Text:
Equal volumes Stopcock closed
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
Pleft 098...View the full answer

Answered By

Leah Muchiri
I am graduate in Bachelor of Actuarial Science and a certified accountant. I am also a prolific writer with six years experience in academic writing. My working principle are being timely and delivering 100% plagiarized free work. I usually present a precised solution to every work am assigned to do. Most of my student earn A++ GRADE using my precised and correct solutions.
4.90+
52+ Reviews
125+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Two 1-L containers at 27 C are connected by a stopcock as pictured below. If each dot in the containers represents 0.0020 mol of a nonreactive gas, what is the pressure in each container before and...
-
Containers A and B in FIGURE CP18.73 hold the same gas. The volume of B is four times the volume of A. The two containers are connected by a thin tube (negligible volume) and a valve that is closed....
-
Two identical vessels are connected by a tube with a valve letting the gas pass from one vessel into the other if the pressure difference p 1.10 atm. Initially there was a vacuum in one vessel while...
-
Suppose a CAP policy was adopted that set allowances for a fixed number of miles driven by each car owner per year. Describe a plan for implementation and trading options.
-
(Multiple choice) (1) If the temperature of an ideal gas is doubled while maintaining constant pressure, the average speed of the molecules (a) Remains constant. (b) Increases by a factor of 4. (c)...
-
1. How comparable are the two different methods? In what ways are they similar? In what ways are they different? 2. What are the positive and negative aspects of each approach that Shocker should...
-
The price of a share is \(\$ 100\). During the following six months the price can go up or down in a \(10 \%\) per month. If the risk-free interest rate is \(8 \%\) per year, continuously compounded,...
-
Under what circumstances would a multiple- priority waiting system be appropriate?
-
Each student will reflect on their results and complete the assignment by posting their answers to the following questions in the Assignment 1 submission folder: Part A- Answer the questions in Part...
-
Northern Tractor is a manufacturer of commercial and consumer garden tractors. In their 30 years of operation Northern has followed a traditional manufacturing process that included maintaining a...
-
Hydrogen gas is frequently prepared in the laboratory by the reaction of zinc metal with sulfuric acid, H 2 SO 4 . The other product of the reaction is zinc(II) sulfate. The hydrogen gas is generally...
-
What is the total pressure, in torr, in a 1.00-L flask that contains 0.0311 mol hydrogen gas collected over water? The temperature is 25 C.
-
Follow the instructions for starting C++ and viewing the SwatTheBugs30.cpp file, which is contained in either the Cpp8\Chap14\SwatTheBugs30 Project folder or the Cpp8\Chap14 folder. (Depending on...
-
A system for recovering methanol from a solid product wet with methanol involves evaporation of the alcohol into a stream of inert gas, essentially nitrogen. In order to recover the methanol from the...
-
The annual return of a risky financial portfolio, denoted by \(R_{p}\), can be described by the following linear regression model: where \(F_{1}\) and \(F_{2}\) are mutually correlated risk factors...
-
Gonzales determined that Company A had an operating cycle of 100 days in 20X2, whereas Company D had an operating cycle of 145 days for the same fiscal year. This means that: A. Company Ds inventory...
-
The most likely tax environment in which Wilson Papers shareholders would prefer that Wilson repurchase its shares (share buybacks) instead of paying dividends is one in which: A. the tax rate on...
-
Consider the following estimated area response model for sugar cane (area of sugar cane planted in thousands of hectares in a region of Bangladesh), as a function of relative price (100 times the...
-
Explain two specific kinds of crime that are on the rise with increasing use of the Internet.
-
A genetically engineered strain of Escherichia coli (E. coli) is used to synthesize human insulin for people suffering from type I diabetes mellitus. In the following simplified reaction scheme,...
-
An electron and a He atom have the same uncertainty in their speed. What can you say about the relative uncertainty in position for the two particles?
-
Describe the trends in Figure 17.5 that you expect to see as the quantum number n increases. Figure 17.5 n-101 -6 -4 -2 k/(1010 n-15 -1.0 -0.5 0.5 1.0 k(1010 m ) n-5 -0.6 -0.4 -0.2 0.2 0.4 0.6...
-
In this problem, we consider the calculations for Ï p and Ï x for the particle in the box shown in Figure 17.5 in more detail. In particular, we want to determine how the absolute...
-
Swifty Company issued $880,000, 9%, 20-year bonds on January 1, 2020, at 102. Interest is payable annually on January 1. Swifty uses the straight-line method of amortization and has a calendar...
-
In the year of her death, P owned a rental property - land (FMV $250,000; cost $200,000), building (FMV $340,000; cost $300,000; UCC $200,000). In her Will, she bequeathed the property to her spouse....
-
The skills of which refers to the practice of confronting an employee with his or her job performance record with the objective of finding ways to overcome deficiencies and improve job performance?

Study smarter with the SolutionInn App


