Use formal charge to predict which of the following skeleton structures is more likely to be correct
Question:
Use formal charge to predict which of the following skeleton structures is more likely to be correct for a molecule with the formula H2CO.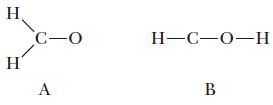
Transcribed Image Text:
H H C-O A H-C-O-H B
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)

Answered By

Somshukla Chakraborty
I have a teaching experience of more than 4 years by now in diverse subjects like History,Geography,Political Science,Sociology,Business Enterprise,Economics,Environmental Management etc.I teach students from classes 9-12 and undergraduate students.I boards I handle are IB,IGCSE, state boards,ICSE, CBSE.I am passionate about teaching.Full satisfaction of the students is my main goal.
I have completed my graduation and master's in history from Jadavpur University Kolkata,India in 2012 and I have completed my B.Ed from the same University in 2013. I have taught in a reputed school of Kolkata (subjects-History,Geography,Civics,Political Science) from 2014-2016.I worked as a guest lecturer of history in a college of Kolkata for 2 years teaching students of 1st ,2nd and 3rd year. I taught Ancient and Modern Indian history there.I have taught in another school in Mohali,Punjab teaching students from classes 9-12.Presently I am working as an online tutor with concept tutors,Bangalore,India(Carve Niche Pvt.Ltd.) for the last 1year and also have been appointed as an online history tutor by Course Hero(California,U.S) and Vidyalai.com(Chennai,India).
4.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
The Understanding section in Example 9.10 showed two possible arrangements for the atoms in H 2 CO. We concluded that structure A, repeated here, was better. A third possible structure follows as B. ...
-
If you were an analytical chemist, one test you might use for the presence of iron(III) ions is to add a solution of potassium thiocyanate, KSCN; a blood-red color indicates that a compound of iron...
-
The sulfate ion, SO 4 2 , is present in a number of important minerals, including gypsum (CaSO 4 2H 2 O), which is used in cement, and Epsom salts (MgSO 4 7H 2 O), which is used as a purgative....
-
16. Read each brief description of a project in the following table and classify its type according to the Obeng model (Fog, Quest, Movie, Paint by Numbers (PBN)). (5) Project Reduce the cost of...
-
If x is in feet, t is in seconds, and v is in feet per second, what are the units of the constants C 1 and C 2 in each part of Problem 6?
-
Briefly explain the operation of process costing, including the manner in which the unit costs of finished goods are determined.
-
Show that the two representations of the Stefan-Maxwell model given by Eqs. (21.4) and (21.5) are equivalent. ns -VA = j=1 y;Ni-yiNj CDij (21.4)
-
An Evaluation of the Dilemma at Benevento Foods: Understanding Cause and Effect and Recommendations for Improvement For this Assignment, you will continue to evaluate the same scenario from the...
-
Given the polynomial: f(x)=-3x - 4x + 2, find the range of polynomial.
-
Describe the main difference between covalent and ionic bonding.
-
Give the characteristics of a correct Lewis structure.
-
What percent of $590 is $950?
-
Which statement describes the measurement of an asset or liability categorized within Level 3 of the fair value hierarchy? It was measured using significant unobservable inputs that reflect market...
-
Stanton Delivery purchased a truck costing $ 1 0 0 , 0 0 0 on September 3 , 2 0 1 7 , by paying $ 4 , 0 0 0 down and signing a 1 0 % , 1 8 0 - day note payable for the balance. Stanton's year end is...
-
At June 1, Wildhorse Co had total assets of $40,350 and total liabilities of $15,500. Calculate each of the following amounts. 1. If total assets increased $12,150 in June and total liabilities...
-
Task Criteria 1. Identify the sources of finance to a company and the reward of the providers of that finance; 2. Discuss the sources and evaluation of risk; 3. Analyse the role of mergers and...
-
link to case : https://1drv.ms/b/s!AloL8CwjfeUZjT5Cfpwgzw3t8Gng?e=KrYA0Q questions : In preparing the case, you can assume that Genentech has 1,052 million shares outstanding. 1. Why is Roche seeking...
-
Mayor George Henry has a problem. He doesnt want to anger voters by taxing them because he wants to be reelected, but the town of Gapville needs more revenue for its schools. He has a choice between...
-
Which should drive action planning more, strengths or weaknesses? That is, is it more important to build on your strengths or to reduce your weaknesses? Explain.
-
Determine whether preparation of each of the following polymers would best be achieved via cationic addition or anionic addition. (a) (b) (c) (d) (e) (f) CN CN CN CN CN CN OMe OMe OMe OMe
-
Arrange the following monomers in order of reactivity toward cationic polymerization. NO2 CH3 OAc
-
Arrange the following monomers in order of reactivity toward anionic polymerization. CI NO2
-
Our teams operate and encounter all types of devices. Provide a brief description of each of the following common network devices: Networking Device Description hub router NIC switch
-
Discuss the availability and security issues of wireless technology. Use suitable business case examples.
-
Using a single diagram or illustration, explain the term encapsulation and the term Protocol Data Unit (PDU). Your explanations should also refer to the terms segments, packets, and frames.

Study smarter with the SolutionInn App


