Use the standard reduction potentials in Table 18.1 To find (a) A metal ion that reduces Ni
Question:
Use the standard reduction potentials in Table 18.1 To find
(a) A metal ion that reduces Ni2+.
(b) A metal ion that can oxidize Cu.
(c) A metal ion that is reduced by Cr2+ but not H2.
Table 18.1 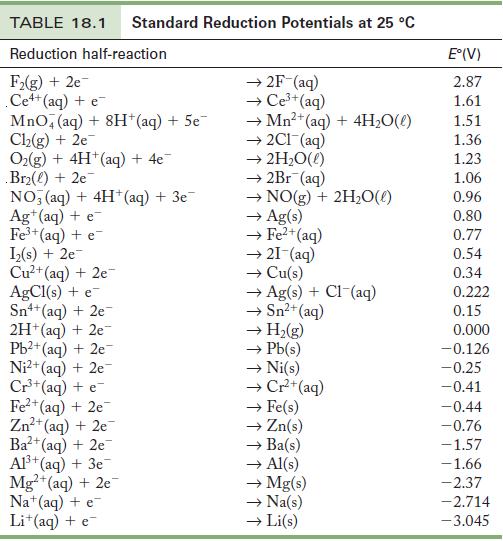
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
To answer these questions we must examine the standard reduction potentials in the table you have pr...View the full answer

Answered By

Pushpinder Singh
Currently, I am PhD scholar with Indian Statistical problem, working in applied statistics and real life data problems. I have done several projects in Statistics especially Time Series data analysis, Regression Techniques.
I am Master in Statistics from Indian Institute of Technology, Kanpur.
I have been teaching students for various University entrance exams and passing grades in Graduation and Post-Graduation.I have expertise in solving problems in Statistics for more than 2 years now.I am a subject expert in Statistics with Assignmentpedia.com.
4.40+
3+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Calculate the potential for each of the voltaic cells in Exercise 18.43 when the concentrations of the soluble species and gas pressures are as follows: Exercise 18.43 Use the standard reduction...
-
Write an expression and determine a value for K eq for each voltaic cell in Exercise 18.43 . Exercise 18.43 Use the standard reduction potentials in Table18.1 to find (a) A metal ion that reduces Ni...
-
Use the standard reduction potentials in Table 18.1 to find (a) A reducing agent that will reduce Cu 2+ but not Pb 2+ . (b) An oxidizing agent that will react with Cu but not Fe 2+ . (c) A metal ion...
-
A magnet of mass 5 . 0 1 kg is suspended from the ceiling by a cord as shown. A large magnet is somewhere off to the right, pulling on the small hanging magnet with a constant force of F = 8 0 . 4 N...
-
A spherical satellite in near-earth orbit is exposed to solar irradiation of 1353 W/m2. To maintain a desired operating temperature, the thermal control engineer intends to use a checker pattern for...
-
Consider building a CSMA/CD network running at 1 Gbps over a 1-km cable with no repeaters. The signal speed in the cable is 200,000 km/sec. What is the minimum frame size?
-
True or False: Belt tension should be very tight. The tighter the better.
-
The Jonesburgh County Basketball Conference (JCBC) is an amateur basketball association. Each city in the county has one team as its representative. Each team has a maximum of 12 players and a...
-
Presented below are selected transactions of Kingbird Company. Kingbird sells in large quantities to other companies and also sell product in a small retail outlet. March 1 Sold merchandise on...
-
Write an expression and determine a value for K eq for each voltaic cell in Exercise 18.44 . Exercise 18.44 Use the standard reduction potentials in Table18.1 to find (a) A reducing agent that will...
-
Use the data in Appendix H and assume standard conditions when answering the following questions.
-
PEI Potatoes processes potatoes into potato cuts at its highly automated plant. For many years, it processed potatoes for only the retail consumer market where it had a superb reputation for quality....
-
Explain the Flower of Service concept and identify each of its petals. What insights does this concept provide for service marketers?
-
What are the dimensions of perceived control, and why is perceived control important?
-
How can a company promote itself through a search engine?
-
Click-through is one measure of the effectiveness of online advertising. a What is click-through? b Which factors are important in determining the click-through rate of a banner advertisement? c Is...
-
How can service firms build brand equity?
-
This problem demonstrates the effects of transactions on the current ratio and the debt ratio of Harrington Company. Harringtons condensed and adapted balance sheet at December 31, 2012, follows....
-
The financial statements of Eastern Platinum Limited (Eastplats) are presented in Appendix A at the end of this textbook. Instructions (a) Does East plats report any investments on its statement of...
-
If water at 180 F is flowing with a velocity of 4.50 ft/s in a standard 6-in Schedule 40 pipe, calculate the weight flow rate in lb/h.
-
The recommended velocity of flow in the discharge line of an oil hydraulic system is in the range of 8.0 to 25.0 ft/s. If the pump delivers 30 gal/min of oil, specify the smallest and largest...
-
Repeat Problem 6.45, except specify suitable sizes for the suction lines to maintain the velocity between 2.0 ft/s and 7.0 ft/s for 30 gal/min of flow. Repeat Problem The recommended velocity of flow...
-
Given the following formula: Monthly Pay= [rate + rate / ([1+rate] ^(months) -1) X principle Where rate of 6% means 6/1200 and Months means number of years x 12 Possible data to test: Principle 12200...
-
What are some features of Cross Laminated Timber?
-
The 201 1 price implied by PPP for the Billy bookcase in the United Kingdom was 137.49, but the actual price at that time was $29.90. What are possible reasons why the price was relatively low?

Study smarter with the SolutionInn App


