What is H rxn for reaction of iron(III) oxide and carbon monoxide to give iron metal and
Question:
What is ΔHrxn for reaction of iron(III) oxide and carbon monoxide to give iron metal and carbon dioxide gas? Use the following reactions: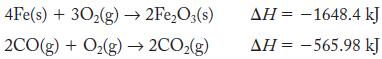
Transcribed Image Text:
4Fe(s) + 30₂(g) → 2Fe₂O3(s) 2CO(g) + O₂(g) → 2CO₂(g) AH-1648.4 kJ ΔΗ = -565.98 kJ
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
To determine Hrxn for the reaction of ironIII oxide and carbon monoxide to give iron metal and carbo...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Iron(III) oxide reacts with carbon monoxide to give iron metal and carbon dioxide. If you begin the reaction with 24.0 g of iron(III) oxide and 34.0 g of carbon monoxide, what is the theoretical...
-
Solid oxide fuel cells (SOFC) have been proposed as an alternative energy technology for use in large stationary power applications (1 to 10MWof electrical power). These devices have an ion...
-
Blast furnaces extract pure iron from the iron(III) oxide in iron ore in a two step sequence. In the first step, carbon and oxygen react to form carbon monoxide: 2C (s) + O 2 (g) 2CO (g) In the...
-
How to respond to this response The effective utilization of data is integral to enhancing efficiency and making informed decisions?
-
An MSMPR-type crystallizer is to be designed to produce 2,000 lb/h of crystals of the heptahydrate of magnesium sulfate with a predominant crystal size of 35 mesh. The magma will be 15 vol% crystals....
-
a. Identify the functions local extreme values in the given domain, and say where they occur. b. Which of the extreme values, if any, are absolute? c. Support your findings with a graphing calculator...
-
Jacobs, Jaohara and Jurd are partners in an importing business of JJJ Partners. The statement of financial position of the partnership as at 30 June 2025 is set out below. Required (a) Prepare the...
-
Data for Schmidt Company is presented in the following table of rates of return on investment and residual incomes: Determine the missing items, identifying each item by the appropriateletter. Income...
-
16. A signal containing multiple frequencies is shown in figure A. It is passed through 2 different filters and then outputs are shown in figure B & C. M (A) S (B) sha (C) (a) B corresponds to low...
-
In a survey of 496 college students, it was determined that 356 owned an iPad. 293 owned a laptop. 285 owned a gaming system. 193 owned an iPad and a laptop. 200 owned an iPad and a gaming system....
-
Ammonium nitrate, a common fertilizer, has been used by terrorists to construct car bombs. The products of the explosion of ammonium nitrate are nitrogen gas, oxygen gas, and water vapor. or ammonium...
-
A typical waterbed measures 84 in. 60 in. 9 in. How many kilocalories are required to heat the water in the waterbed from 55 F (cold water from the faucet) to 85 F, the operating temperature of the...
-
If possible, find the slope of the line pass- ing through each pair of points. (-). (- -)
-
Two masses \(m_{1}=5.0 \mathrm{~kg}\) and \(m_{2}=2.0 \mathrm{~kg}\) are joined by an inextensible rope of negligible mass. The mass \(m_{1}\) rests on a rough horizontal plane while \(m_{2}\) is...
-
A particle \(\mathrm{P}\) is moving on a circumference of radius \(R=3.50 \mathrm{~m}\) according to an angular speed given by \(\omega(t)=k t^{2}\), with \(k=13.18\) degrees \(/ \mathrm{s}^{3}\) and...
-
The eminent sociologist Zygmunt Bauman had this to say about social media: Most people use social media not to unite, not to open their horizons wider, but on the contrary, to cut themselves a...
-
A body of mass \(m=2.4 \mathrm{~kg}\) slides on a rough horizontal plane, with static and dynamic coefficients of friction \(\mu_{s}=0.45\) and \(\mu_{d}=0.35\), respectively. If the body is...
-
A passenger car is traveling at \(v_{0}=11.1 \mathrm{~m} / \mathrm{s}\) downhill along a road of slope \(15^{\circ}\). At a certain instant it brakes by locking all wheels simultaneously until it...
-
How does the PV of depreciation tax shields vary across the recovery-period classes shown in Table? Give a general answer; then check it by calculating the PVs of depreciation tax shields in the...
-
Jax Incorporated reports the following data for its only product. The company had no beginning finished goods inventory and it uses absorption costing. $ 57.30 per unit $ 10.30 per unit $ 7.80 per...
-
In the description of Figure 9.24b, the following sentence appears: At the point when the L 2 phase disappears, the temperature increases beyond 94°C and the vapor composition changes along the i...
-
Explain why chemists doing quantitative work using liquid solutions prefer to express concentration in terms of molality rather than molarity.
-
Explain the usefulness of a tie line on a PZ phase diagram such as that of Figure 9.4. Figure 9.4 100 Liquid 80 60 Liquid + vapor 40 Vapor 20 0.2 0.4 0.6 0.8 Zbenzene Pressure/Torr
-
Timeless Toys purchases a product from overseas, including insurance and shipping costs, for $24 per unit. Timeless marks the toy up by 40% to $33.60. Other traceable direct costs amount to $2.90 per...
-
Tootsie Roll Industries has two business segments, one for operations in the United States and one for operations in Mexico and Canada. The following information (in thousands) comes from a recent...
-
What is true when developing rules for organizational codes of conduct?

Study smarter with the SolutionInn App


