Which of the following reactions are formation reactions? Strategy Look for reactions that have all elements in
Question:
Which of the following reactions are formation reactions?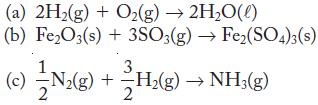
Strategy
Look for reactions that have all elements in their standard states as reactants and one mole of a compound as a product.
Transcribed Image Text:
(a) 2H₂(g) + O₂(g) → 2H₂O(1) (b) Fe₂O3(s) + 3SO3(g) → Fe₂(SO4)3(s) (c) N₂(g) + H₂(g) → NH₂(g) = 3. 2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
a No this is not a proper formation reaction because two ...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
The standard heats of formation of H2O(g), H2S(g), H2Se(g), and H2Te(g) are -241.8, -20.17, +29.7, and +99.6 kJ/mol, respectively. The enthalpies necessary to convert the elements in their standard...
-
State the value of the padding field in SHA-512 if the length of the message is: a). 1919 bits, b). 1920 bits, c). 1921 bits.
-
Which of the following reactions are possible, and by what interaction could they occur? For those forbiddevn explain why. (a) + p K0 + p + 0 (b) K + p 0 + 0 (c) K+ + n + + 0 + y (d) K + 0 + 0 + +...
-
Local 54 has retained James Love to represent it in a grievance against Dilated Peoples Optical Inc. The union is grieving the employers decision to exclude from the bargaining unit the position of...
-
Silver ion in methanol was exchanged with sodium ion using Dowex 50 cross-linked with 8% divinyl benzene by Gable and Stroebel [J. Phys. Chem., 60, 513-517 (1956)]. The molar selectivity coefficient...
-
A talk on the dangers of high cholesterol was given to eight workers. Each workers cholesterol was tested both before the talk and several weeks after the talk, with the results given below. What is...
-
Suppose Emory rejects the lease we analyzed, and the lessor agrees to lower the lease rate to $11,700 per year. Does this change make the lease attractive?
-
Zero beginning inventory, materials introduced in middle of process. Roary Chemicals has a Mixing Department and a Refining Department. Its process-costing system in the Mixing Department has two...
-
One key limitation of Sage 50 Accounting software, while there is a Sage 50 cloud version that offers some cloud-based features, the mobile access and functionality may not be as extensive as with...
-
A convenience store is considering changing its layout to encourage impulse buying. The triangular flow matrix below gives the measure of association between different product groups ( e. g., beer,...
-
One step in the production of nitric acid, a powerful acid used in the production of fertilizers and explosives, is the combustion of ammonia. Use Equation 5.10, with the enthalpies of formation of...
-
Which of the following reactions are formation reactions? (a) H(g) + O(g) HO(g) (b) Fe(s) + N(g) + 302(g) Fe(NO3)2(s) 3. (c)NH(g) N(g) + H(g) 2 2
-
An investment earned 6% compounded semiannually for two years and 8% compounded annually for the next three years. What was the equivalent annually compounded rate of return for the entire five-year...
-
Solicitation occurs when a person entices, advises, incites, orders, or otherwise encourages someone else to commit a crime. For solicitation, there is no requirement that the crime actually be...
-
If you use an extension cord, current travels from the 120 V outlet, along one wire inside the cord, through the appliance youve plugged into the cord, and back to the outlet through a second wire in...
-
Sleepy Recliner Chairs completed the following selected transactions: Record the transactions in the journal of Sleepy Recliner Chairs. Explanations are not required. (Round to the nearest dollar.)...
-
A weak mixture of propane \(\left(\mathrm{C}_{3} \mathrm{H}_{8} ight)\) and \(50 \%\) excess air is ignited in a constant volume combustion chamber. The initial conditions were \(1 \mathrm{bar}\) and...
-
In his book, The Post-Industrial Society, Peter Drucker wrote: Society, community, and family are all conserving institutions. They try to maintain stability and to prevent, or at least slow down,...
-
In Section 10-4 we considered two production technologies for a new Wankel-engined outboard motor. Technology A was the most efficient but had no salvage value if the new outboards failed to sell....
-
Compile data on consumption and expenditures for the following categories in 30 different countries: (1) food and beverages, (2) clothing and footwear, (3) housing and home operations, (4) household...
-
Propose an efficient synthesis for each of the following compounds using the malonic ester synthesis. (a) (b) (c) (d) (e)
-
Starting with diethyl malonate and using any other reagents of your choice, proposean efficient synthesis for each of the following compounds: (a) (b) (c) .
-
The malonic ester synthesis cannot be used to make 2,2-dimethylhexanoic acid. Explain why not.
-
Using the costs for January through December below, calculate the following: a. b. C. Estimate the company's variable cost per unit. Estimate the company's total fixed costs. Estimate the company's...
-
The binomial 2-49 has y - 7 as one of its factors. What is the other factor of the binomial?
-
MAC4865 JANUARY/FEBRUARY 2024 QUESTION 3 (35 marks - 63 minutes) Question 3 consists of 2 independent parts. Part A Ipuleti (Pty) Ltd have two wholly owned subsidiaries, Senoelo (Pty) Ltd and Khaba...

Study smarter with the SolutionInn App


