Which of the following reactions are formation reactions? (a) H(g) + O(g) HO(g) (b) Fe(s) +
Question:
Which of the following reactions are formation reactions?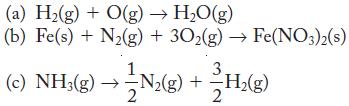
Transcribed Image Text:
(a) H₂(g) + O(g) → H₂O(g) (b) Fe(s) + N₂(g) + 302(g) → Fe(NO3)2(s) 3. (c)NH₂(g) →N₂(g) + H₂(g) 2 2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
a N...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
a. Which of the following reactions would lead to the formation of an amide? 1. 2. 3. 4. 5. 6. b. For those reactions that do form amides, what could you do to improve either the rate of amide...
-
Which of the following reactions are possible, and by what interaction could they occur? For those forbiddevn explain why. (a) + p K0 + p + 0 (b) K + p 0 + 0 (c) K+ + n + + 0 + y (d) K + 0 + 0 + +...
-
The firm is Harvest Clean Eats which is located in Halifax, Nova Scotia Please discuss their target key Competitors?
-
The following are selected items from the accounting records of Georgia Peach for the year ended December 31, 2015: Note payable to SmithField Bank . . . . . . . . . . . . . . . . . . . . . . . . . ....
-
Derive (15-44). Use this equation to solve the following problem. Sulfate ion is to be removed from 60 L of water by exchanging it with chloride ion on 1 L of a strong-base resin with relative molar...
-
At the beginning of 2015, you have $100 invested in a stock. Each year there is a 60 percent chance that the stock increases 50 percent, a 20 percent chance the stock goes up 10 percent, and a 20...
-
Consider a seven-year fair market value lease for a \($12.5\) million Gulfstream jet with a remaining useful life of 10 years. Suppose the monthly lease payments are \($175,000\) and the appropriate...
-
Selected data for Richmond Company for 2011 and additional information on industry averages follow. Required a. Calculate and compare Richmond Company's ratios with the industry averages. b. Discuss...
-
Other than the wind speed, what factor has the most impact on the amount of power generated by a wind turbine? Swept area of rotor Turbine style Terrain Blade material.
-
It is October 16, 2020, and you have just taken over the accounting work of China Moon Products, whose annual accounting period ends October 31. The company?s previous accountant journalized its...
-
Which of the following reactions are formation reactions? Strategy Look for reactions that have all elements in their standard states as reactants and one mole of a compound as a product. (a) 2H(g) +...
-
What characteristic does every exothermic reaction have?
-
The line graph shows the cost of inflation. What cost $10,000 in 1984 would cost the amount shown by the graph in subsequent years. Here are two mathematical models for the data shown by the graph....
-
(a) One of the main problems encountered in the design of a diesel engine combustion system is the mixing of the air and fuel sufficiently rapidly to ensure complete combustion. Explain, using...
-
An electric cell is based on the reaction \(\mathrm{Pb}+\mathrm{Hg}_{2} \mathrm{Cl}_{2} ightarrow \mathrm{PbCl}_{2}+2 \mathrm{Hg}\). If the enthalpy of reaction for this reaction, \(Q_{p}\), at...
-
Calculate the emf of a hydrogen-oxygen fuel cell operating reversibly if the overall reduction in Gibbs energy is \(238 \mathrm{MJ} / \mathrm{kg} \mathrm{H}_{2}\). If the cell operates at \(75 \%\)...
-
The exhaust gases of a compression-ignition engine are to be used to drive an exhaust gas turbo-supercharger. Estimate the mean pressure ratio of expansion and the isentropic enthalpy drop per kmol...
-
According to many financial experts, the logical place to begin the search for quality investments is to examine the products and services you use on a regular basis. This suggestion is based on the...
-
Describe each of the following situations in the language of options: a. Drilling rights to undeveloped heavy crude oil in Northern Alberta. Development and production of the oil is a negative-NPV...
-
The Adjusted Trial Balance columns of a 10-column work sheet for Webber Co. follow. Complete the work sheet by extending the account balances into the appropriate financial statement columns and by...
-
When a malonic ester synthesis is performed using excess base and 1,4-dibromobutane as the alkyl halide, an intramolecular reaction occurs, and the product contains a ring. Draw the product of this...
-
Starting with ethyl acetoacetate and using any other reagents of your choice, propose an efficient synthesis for each of the following compounds. (a) (b) (c) OH
-
The acetoacetic ester synthesis cannot be used to make 3,3-dimethyl-2-hexanone. Explain why not.
-
Instructions: You are to place the items below in their proper place in the statement of cash flows and also decide whether each item should be a debit or credit Your statement of cash flows will be...
-
STATES OF NATURE MACHINE Very High Moderate Low High R 25 15 10 20 S 20 12 25 15 T 40 30 -5 -20 *The values are in thousand pesos. What is the best alternative if it is known that the sales will be...
-
what ways does the study of phenotypes inform our understanding of complex biological phenomena, including disease susceptibility, ecosystem dynamics, and evolutionary processes, providing insights...

Study smarter with the SolutionInn App


