Write the formation reaction for each of the following substances. (a) HBr(g) (b) HSO4(l) (c) 03(g) (d)
Question:
Write the formation reaction for each of the following substances.
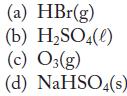
Transcribed Image Text:
(a) HBr(g) (b) H₂SO4(l) (c) 03(g) (d) NaHSO4(s)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (2 reviews)
a b c...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
The following data compare the standard enthalpies and free energies of formation of some crystalline ionic substances and aqueous solutions of the substances: (a) Write the formation reaction for...
-
Write a proper formation reaction for each substance. a. O3(g) b. Na2O2(s) c. PCl5(g)
-
Write a proper formation reaction for each substance. a. H3PO4(s) b. Na2O(s) c. C3H7OH()
-
In the figure below, which compares different DNA extraction kits (e.g, MO BIO and PSP), which one of the two trees shows the co-occurrence of microbial taxa? Proportion of max 0 0.2 0.4 0.6 0.8...
-
Using the screen analysis of Exercise 17.3, calculate, with a spreadsheet, the surface-mean, mass-mean, arithmetic-mean, and volume-mean crystal diameters, assuming that all particles have the same...
-
Find the most general antiderivative or indefinite integral. You may need to try a solution and then adjust your guess. Check your answers by differentiation. [2 2x(1-x-) dx
-
Adamson and Baker formed a partnership by investing \($220\) 000 and \($180\) 000 respectively. The partnership had a final profit of \($92\) 000 in the first year. Required a. Prepare the journal...
-
The following cumulative frequency polygon shows the selling price ($000) of houses sold in the Billings, Montana, area. a. How many homes were studied? b. What is the class interval? c. One hundred...
-
7. The full symbol of an ion of an element is 39X+? Which one of the following is the number of electrons in the ion? A. 18 B. 19 C. 20 D. 39 8. The atomic numbers of elements W, X, Y and Z are 9,...
-
The Evanstonian is an upscale independent hotel that caters to both business and leisure travelers. When a guest calls room service at The Evanstonian, the room-service manager takes down the order....
-
Write the chemical equation for the reaction whose energy change is the standard enthalpy of formation of each of the following substances. (a) CHCOOH(e) (b) HPO4(0) (c) CaSO4 2HO(s) (d) C(s, diamond)
-
What does an energy-level diagram for the reverse reaction from Exercise 5.72 look like? Exercise 5.72 In the process of isolating iron from its ores, carbon monoxide reacts with iron(III) oxide, as...
-
The author currently has seven different books in print. If those seven books are stacked in a random order, what is the probability that they are arranged in alphabetical order from top to bottom?...
-
Closed cycle gas turbines operate on the internally reversible Joule cycle with an efficiency of \[\eta_{\text {Joule }}=1-\frac{1}{r_{p}^{(\kappa-1) / \kappa}}\] where \(r_{p}=\) pressure ratio of...
-
Explain why the Carnot cycle efficiency is unrealistically high for a real engine. Introducing the concept of external irreversibility, evaluate the efficiency of an endoreversible engine at maximum...
-
Predict the population of your city or state for the year 2020.
-
Problem P3.12 seems to demonstrate that the efficiency of the reheated Rankine cycle gets better as the work distribution between the high pressure (HP) and low pressure (LP) turbines becomes more...
-
A heat engine operates between two finite reservoirs, initially at 800 and \(200 \mathrm{~K}\), respectively. The temperature of the hot reservoir falls by \(1 \mathrm{~K}\) for each \(1...
-
Consider three investors: a. Mr. Single invests for one year. b. Ms. Double invests for two years. c. Mrs. Triple invests for three years. Company Zs earnings and dividends per share are expected to...
-
Experiment: Tossing four coins Event: Getting three heads Identify the sample space of the probability experiment and determine the number of outcomes in the event. Draw a tree diagram when...
-
Discuss how the DebyeHckel screening length changes as the (a) Temperature (b) Dielectric constant, (c) Ionic strength of an electrolyte solution are increased.
-
Why is it not possible to measure the Gibbs energy of solvation of Cl directly?
-
Why are activity coefficients calculated using the DebyeHckel limiting law always less than one?
-
On May 10, 2016, Oracle stock traded for about $120. All things equal, if Oracle had never had a stock split, what would a share of Oracle have traded for that same day? Round answer to the nearest...
-
Now that you know a bit more about mortgages, we can look at how to apply this information. Knowing how to calculator mortgage payments, amortization schedules and paying attention to property taxes...
-
The Manager at Deli Depot believes that Food Quality is an important driver of customer satisfaction. A Pearson correlation of the two variables reveals the following: Correlations X13-- Satisfaction...

Study smarter with the SolutionInn App


