Shown here are three pairs of hybrid orbitals, with each set at a characteristic angle. For each
Question:
Shown here are three pairs of hybrid orbitals, with each set at a characteristic angle. For each pair, determine the type of hybridization, if any, that could lead to hybridorbitals at the specified angle.
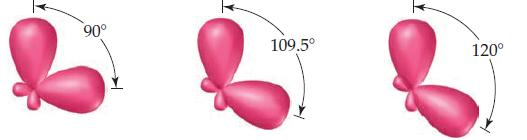
Transcribed Image Text:
90° 109.5° 120°
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
90 degree angles are formed by d 2 sp 3 orbitals where there is an ...View the full answer

Answered By

Muhammad Ahtsham Shabbir
I am a professional freelance writer with more than 7 years’ experience in academic writing. I have a Bachelor`s Degree in Commerce and Master's Degree in Computer Science. I can provide my services in various subjects.
I have professional excellent skills in Microsoft ® Office packages such as Microsoft ® Word, Microsoft ® Excel, and Microsoft ® PowerPoint. Moreover, I have excellent research skills and outstanding analytical and critical thinking skills; a combination that I apply in every paper I handle.
I am conversant with the various citation styles, among them; APA, MLA, Chicago, Havard, and AMA. I also strive to deliver the best to my clients and in a timely manner.My work is always 100% original. I honestly understand the concern of plagiarism and its consequences. As such, I ensure that I check the assignment for any plagiarism before submission.
4.80+
392+ Reviews
587+ Question Solved
Related Book For 

Chemistry The Central Science
ISBN: 9780321910417
13th Edition
Authors: Theodore E. Brown, H. Eugene LeMay, Bruce E. Bursten, Catherine Murphy, Patrick Woodward, Matthew E. Stoltzfus
Question Posted:
Students also viewed these Sciences questions
-
List causes that could lead to a variable overhead spending variance?
-
List causes that could lead to a variable overhead efficiency variance?
-
List causes that could lead to a fixed overhead spending variance?
-
The distance between the K+ and Cl ions in KCl is 2.80 1010 m. Calculate the energy required to separate the two ions to an infinite distance apart, assuming them to be point charges initially at...
-
Determine whether the ordered pair (1 / 2, 9 / 10) is a solution of the equation 5y - 4 = x.
-
Explain the principles of numerical taxonomy and the algorithms utilized in clustering analysis to classify organisms based on quantitative characters.
-
Define project management principles in the context of software development.
-
The total wages and salaries earned by all employees of Langen Electronics, Ltd. during March, as shown in the labor cost summary and the schedule of fixed administrative and sales salaries, are...
-
Alcoa's offer to Reynolds Metals consisted of $4.3 billion in cash plus the assumption of $1.5 billion in Reynolds' outstanding debt. Alcoa's offer letter, which it made public, from its chief...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
(a) Consider the AF 3 molecules in Exercise 9.27. Which of these will have a nonzero dipole moment? (b) Which of the AF 4 molecules in Exercise 9.28 will have a zero dipole moment?
-
Consider the Lewis structure for glycine, the simplest amino acid: (a) What are the approximate bond angles about each of the two carbon atoms, and what are the hybridizations of the orbitals on each...
-
Use an electronic calculator equipped with a natural logarithm function to evaluate each of the following. 1.01-120 In 850 0.01
-
Explore each of the following resources below. What did you learn? How might each be helpful in the start-up of a business? Maintenance? Dissolution? Useful Online Legal Resources Business.gov...
-
(a) Perform the following unit conversions: a. 1 L to in 3 b. 650 J to Btu c. 0.135 kW to ft lbf/s d. 378 g/s to lb/min. e. 304 kPa to lbf/in.2 f. 55 m/h to ft /s g. 50 km/h to ft/s h. 8896 N to ton...
-
It is March 1, 2021. You work as a financial analyst for Umbrella Corporation in Trenton, New Jersey. Dr. James Marcus, CEO, has arranged for the purchase of a British gene sequencing system. The...
-
For our discussion forum we are going to comment on a short video found in the website entitled "The truth about algorithms" by Cathy O'Neil https://www.youtube.com/watch?v=heQzqX35c9A 1. What is the...
-
Watch the biological legal drama film titled Erin Brockovich. Make an essay composed of 500 to 1000 words. Please follow these guide questions to help you create your essay: 1. In your perspective...
-
The file DomesticBeer contains the percentage alcohol, number of calories per 12 ounces, and number of carbohydrates (in grams) per 12 ounces for 158 of the best-selling domestic beers in the United...
-
Prepare a stock card using the following information A company is registered for GST which it pays quarterly, assume GST was last paid on the 30th of June 2019. It uses weighted average cost...
-
Imagine a container placed in a tub of water, as depicted in the accompanying diagram. (a) If the contents of the container are the system and heat is able to flow through the container walls, what...
-
The automobile fuel called E85 consists of 85% ethanol and 15% gasoline. E85 can be used in so-called "flex-fuel" vehicles (FFVs), which can use gasoline, ethanol, or a mix as fuels. Assume that...
-
At 20C (approximately room temperature) the average velocity of N2 molecules in air is 1050 mph. (a) What is the average speed in m/s? (b) What is the kinetic energy (in J) of an N2 molecule moving...
-
All scheduled presentations and events will take place in the Main Conference Room, 1st Floor. Time February 25 Registration Guide to the Job Fair Rsum Writing Lunch (refreshments served) The...
-
You have work experience in the human resource management field. You want to further understand what factors make a project successful. You are particularly interested in employee selection and...
-
How many defects must a unit have in order to be defined as a defective unit?

Study smarter with the SolutionInn App


