Which arrangement of cations (yellow) and anions (blue) in a lattice is the more stable? Explain your
Question:
Which arrangement of cations (yellow) and anions (blue) in a lattice is the more stable? Explain your reasoning.
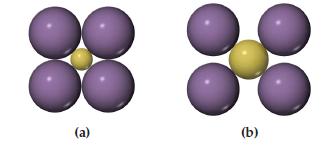
Transcribed Image Text:
(a) (b)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
Recall that similar charges have strong repulsive tendencies against ea...View the full answer

Answered By

Vijesh J
My passion to become a tutor is a lifetime milestone. Being a finance and marketing professional with hands-on experience in wealth management, portfolio management, team handling and actively contributing in promoting the company. Highly talented in managing and educating students in most attractive ways were students get involved. I will always give perfection to my works. Time is the most important for the works and I provide every answer on time without a delay. I will proofread each and every work and will deliver a with more perfection.
4.70+
5+ Reviews
15+ Question Solved
Related Book For 

Chemistry The Central Science
ISBN: 9780321910417
13th Edition
Authors: Theodore E. Brown, H. Eugene LeMay, Bruce E. Bursten, Catherine Murphy, Patrick Woodward, Matthew E. Stoltzfus
Question Posted:
Students also viewed these Sciences questions
-
Explain why anions are always larger than the atoms from which they are derived, whereas cations are always smaller than the atoms from which they are derived.
-
Consider the two stereo isomers of 3-isoproplcyclohexanol. (a) Which is the more stable conformation of each stereo isomer? (b) Which is the more stable stereo isomer by how much?
-
Name the following compound, identify each substituent as axial or equatorial, and tell whether the conformation shown is the more stable or less stable chair form (yellow-greenC1):
-
Accounts Receivable AED 5,000 Accounts Payable 15,000 Advertising Expense 2,000 16,500 Building Cash Common Stock 50,000 Dividends 1,200 Equipment 2,000 Land 70,000 Notes Payable 60,000 3,400 Office...
-
Oxygen at 100 kPa and 200oC is compressed to half its initial volume. Determine the final state in terms of pressure (p2) and temperature (T2) if the compression is carried out in an (a) Isobaric....
-
Davita Company has estimated its yearly overhead cost at $ 1 , 2 0 0 , 0 0 0 and its yearly factory labor cost at $ 4 0 0 , 0 0 0 . It has been determined that the best cost driver for overhead...
-
Records show that the probability is 0.00008 that a truck will have an accident on a certain highway. Use the formula for the Poisson distribution to approximate the probability that at least 5 of...
-
Was Jay Cohens conviction justified?
-
Term Debt The Company has outstanding Notes, which are senior unsecured obligations with interest payable in arrears. The following table provides a summary of the Company's term debt as of September...
-
Complete Keith's tax return including all required schedules and forms using prince edward island as province, using the fillable forms package. Taxpayers Information Taxpayer #1 Name: Keith Dox...
-
The electronic structure of a doped semiconductor is shown here. (a) Which band, A or B, is the valence band? (b) Which band is the conduction band? (c) Which band consists of bonding molecular...
-
Two solids are shown below. One is a semiconductor and one is an insulator. Which one is which? Explain your reasoning.
-
The compound potassium sesquoxide has the empirical formula K 2 O 3 . Show that this compound can be described by an appropriate combination of potassium, peroxide, and superoxide ions. Write a Lewis...
-
After reviewing the annual report from Unilever. Show how Unilever is responding to the digital revolution and other major changes in the business environment with a new marketing model. In addition,...
-
The continuously compounded yield curve is y(T) = 0.04 0.03e-0.47 (a) What is the yield on a zero-coupon bond that matures in 3 months? Express your answer as a percent, to the nearest basis point....
-
Year Amount ($) 1 R 2000 2 2,200 RM 3000 3 3,000 RM 4000 An investment made today will pays you the above cash flows at the end of each year. If your required rate of return is 5% annual interest,...
-
Ensure your response is positive and appropriate and that the children will learn about how their actions affect others without feeling ashamed or humiliated Susan(4yrs 2mnths) and Rachel (4yrs...
-
Identify the different group dynamics as it pertains to the business organizational model (i.e., hidden agendas, giving direction, and using exercises). Why is it important for you to understand the...
-
What is the limitation on a deductible IRA contribution for 2014?
-
A heat engine has a heat input of 3 Ã 104 Btu/h and a thermal efficiency of 40 percent. Calculate the power it will produce, in hp. Source 3 x 10 Btu/h 40% HE Sink
-
How does a trigonal pyramid differ from a tetrahedron so far as molecular geometry is concerned?
-
Describe the bond angles to be found in each of the following molecular structures: (a) Planar trigonal, (b) Tetrahedral, (c) Octahedral, (d) Linear.
-
(a) What is meant by the term electron domain? (b) Explain in what way electron domains behave like the balloons in Figure 9.5.Why do they do so? Two balloons linear orientation Three balloons...
-
Scenario: Yarning Circle and Talking Stick Sarah has recently been employed by Blue Bay Early Learning Centre. Sarah is very proud of having just achieved the Diploma of Children's Services and she...
-
The principal at your school decides to retire, and several well-qualified teachers on the staff apply for the position. Mr. England has been teaching for four years and has his administrator's...
-
Sandi's parents have been divorced since she was five years old. She is now seven. She lives with her mother and visits her father every other weekend. He brings her to school on Monday mornings...

Study smarter with the SolutionInn App


