A soft x-ray photon with an energy of 41.8 eV is absorbed by a hydrogen atom in
Question:
A "soft x-ray" photon with an energy of 41.8 eV is absorbed by a hydrogen atom in its ground state, knocking the atom's electron out. What is the speed of the electron as it leaves the atom?
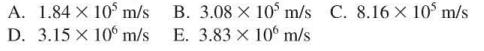
Transcribed Image Text:
A. 1.84 x 105 m/s D. 3.15 x 10 m/s B. 3.08 x 10 m/s C. 8.16 x 105 m/s E. 3.83 x 10 m/s
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Energy of the absorbed photon Ephoton 418 eV Ionization energy E ionization for h...View the full answer

Answered By

Omar ELmoursi
I'm Omar, I have Bachelor degree in Business and Finance, My unique approach is to help students with questions and assignments, I can teach Business, Math, Accounting, Managerial Accounting, Economy, Human resources management, organizational behavior, project management, I have experience dealing with different types of students and teach them how to deal with different types of exercises.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

College Physics A Strategic Approach
ISBN: 9780321595492
2nd Edition
Authors: Randall D. Knight, Brian Jones, Stuart Field
Question Posted:
Students also viewed these Physics questions
-
The energy-level scheme for the hypothetical one electron element Searsium is shown in Fig. E39.25. The potential energy is taken to be zero for an electron at an infinite distance from the nucleus....
-
A hydrogen atom in its ground state absorbs a 97-nm ultraviolet photon. It then emits one or more photons to return to the ground state. (a) If the atom is at rest before absorbing the UV photon,...
-
2.Complete the appropriate answer boxes online: Supply Nature of Supply Key word that determines the Place of Supply 1 Sale of Quick Tax 2 Franchise arrangement 3 Sale of building 4 Legal services 5...
-
Why is it so difficult to assess whether a firm is properly valued?
-
Determine the amount to be paid in full settlement of each of the following invoices, assuming that credit for returns and allowances was received prior to payment and that all invoices were paid...
-
Go to the U.S. Department of Housing and Urban Development (HUD) Web site at www.hud.gov and identify the three topics under Homes that are of interest to you. For each of the three topics, tell...
-
When the Concepcions went to AT&T to get a phone, they were confused as to why they were paying money for the phone when it was advertised as being free. They had to pay sales tax on the full normal...
-
The issue had come up again and again in various management meetings and company seminars. Novartis had too many products and needed to reduce the product proliferation that had occurred. Thomas...
-
What is the relationship between voltage drop and the resistance in the circuit? Explain.
-
Atom A has a larger atomic mass than atom B. Does this mean that atom A also has a larger atomic number? Explain.
-
A sample contains a mix of isotopes of an element. Using a spectrometer to measure the spectrum of emitted light will not reveal the mix of isotopes; analyzing the sample with a mass spectrometer...
-
Why might it be necessary for a parent company to prepare unconsolidated financial statements as well as consolidated statements?
-
A fermentation process involving a single stage chemostat is used to produce the amino acid lysine. The same process has been modified to include cell recycle, with a recycle ratio of 0.3 and a cell...
-
Discuss the various reasons behind Demand and supply conditions differ among countries?
-
On January 1, 2017, Babb Trailers, Inc. issued $6,500,000 of par value bonds for $6,200,000. The bonds pay interest semiannually on January 1 and July 1. The contract rate of interest is 3% while the...
-
Do we have an obligation to help other people? Why or why not?
-
A firm orders apparels from a distant country and can order only once for the entire season. This ordering follows the assumption of the newsvendor model. The firm forecasts that their average demand...
-
For many years New York Studios has produced television shows and operated several FM radio stations. Late in the current year, the radio stations were sold to Times Publishing, Inc. Also during the...
-
The sales department of P. Gillen Manufacturing Company has forecast sales in March to be 20,000 units. Additional information follows: Finished goods inventory, March 1 . . . . . . . . . . . . . . ....
-
FIGURE EX16.8 is a picture at t = 0 s of the particles in a medium as a longitudinal wave is passing through. The equilibrium spacing between the particles is 1.0 cm. Draw the snapshot graph D(x, t =...
-
FIGURE Q16.9 shows the wave fronts of a circular wave. What is the phase difference between (a) Points A and B (b) Points C and D (c) Points E and F? D E A'
-
FIGURE EX16.9 is the snapshot graph at t = 0 s of a longitudinal wave. Draw the corresponding picture of the particle positions, as was done in Figure 16.9b. Let the equilibrium spacing between the...
-
Cam corp. is a manufacturer of wholesale goods. It currently sells to two products. Product A priced at 2,000 and B priced at 1,000. Cam Corp is trying to understand the true profit for each product...
-
Are there penalties for filing the tax return late? If yes, what is the penalty? Services plans to sell the equipment it owns on January 1, 2024. What is the gain from the sale of the equipment If...
-
U.S. Clorox experienced a surge in demand for these products during the pandemic, which lead to a 15.0% increase in sales volume during fiscal 2020. In total, the segment's sales growth for 2020...

Study smarter with the SolutionInn App


