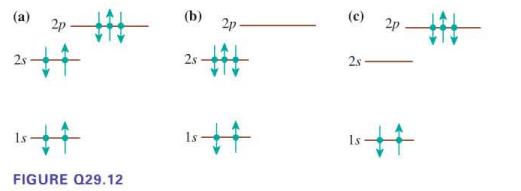
Does each diagram in Figure Q29.12 represent a possible electron configuration of a neutral element? If so,
Question:
Does each diagram in Figure Q29.12 represent a possible electron configuration of a neutral element? If so,
(i) Identify the element
(ii) Determine if this is the ground state or an excited state. If not, why not?
Transcribed Image Text:
(a) 2p 25+4 FIGURE Q29.12 (b) 2s 2p. # (c) 2s 2p 15 #
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
Yes each diagram in Figure Q2912 represents a possible electron configuration of a neutral element L...View the full answer

Answered By

Jayshree Rathi
Hello Students!
This is Jayshree Rathi. I work on a number of renowned student-centric channels such as Chegg, coursehero, as a certified private tutor.
If you are looking for relevant and original content to complete your assignments, essays, and homework, then contact me and within the promised time, I will deliver you your personalized academic work and help you score the best.
4.80+
1+ Reviews
10+ Question Solved
Related Book For 

College Physics A Strategic Approach
ISBN: 9780321595492
2nd Edition
Authors: Randall D. Knight, Brian Jones, Stuart Field
Question Posted:
Students also viewed these Physics questions
-
Does each of the configurations in Figure Q 41.6 represent a possible electron configuration of an element? If so, (i) identify the element and (ii) determine whether this is the ground state or an...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Identify the element for each of these electron configurations. Then determine whether this configuration is the ground state or an excited state. a. 1s 2 2s 2 2p 5 3s b. 1s 2 2s 2 2p 6 3s 2 3p 6 4s...
-
Assume that your team has been in contract with the headquarters of a company that owns several restaurants in different states in the US. Your team is to provide software that manages these...
-
Which financial institution is usually the most important to businesses?
-
On January 1, 20X2, The GenKota Winery purchased a new bottling system. The system has an expected life of 5 years. The system cost \($325\),000. Shipping, installation, and set up was an additional...
-
It would be helpful to me if I could start by asking you to basically tell me about your job. Okay?
-
For the past several years, Shane Banovich has operated a part-time consulting business from his home. As of October 1, 2012, Shene decided to move to rented quarters and to operate the business as...
-
What is one of the Oracle database upgrade methods. Which of the methods offers the least control over the upgrade process? Please explain thoroughly because I am having a hard time with this subject...
-
A researcher observes hydrogen emitting photons of energy 1.89 eV. What are the quantum numbers of the two states involved in the transition that emits these photons?
-
For a hydrogen atom, list all possible states (n, l, m, m.) that have E = -1.51 eV.
-
PLX Refinery Co is a large oil refinery business in Kayland. Kayland is a developing country with a large and growing oil exploration and production business which supplies PLX with crude oil....
-
The difference of specific heats for an ideal gas, \(c_{p, m}-c_{v, m}=\Re\). Evaluate the difference in specific heats for gases obeying (1) the van der Waals and (2) the Dieterici equations of...
-
A single fair die is rolled. Let the event \(A\) be the face showing is even. Let the event \(B\) be the face showing is divisible by 3 . (a) List out the sample space of the experiment. (b) List the...
-
Let \(X\) be a random variable having probability density function \[ f(x)=2 x \text { for } \quad 0 \quad x \quad 1 \] (a) Find \(P\left(\begin{array}{ll}X \quad 75\end{array} ight)\). (b) Find...
-
Let \(X\) have the uniform distribution. (a) Find \(\mathrm{E}[X]\). (b) Find \(\operatorname{Var}[X]\). (c) Find \(P\left(\begin{array}{ll}X \quad 25\end{array} ight)\). (d) Find \(P(33
-
Let \(X\) have a \(\operatorname{beta}(12\) 4) distribution. (a) Find \(\mathrm{E}[X]\). (b) Find \(\operatorname{Var}[X]\).
-
Carol Co. traded in an old machine costing $22,000 for a new machine for a cash price of $18,000 with a trade-in allowance of $7,000. Accumulated Depreciation on the old machine was $14,000. a. What...
-
A consultant is beginning work on three projects. The expected profits from these projects are $50,000, $72,000, and $40,000. The associated standard deviations are $10,000, $12,000, and $9,000....
-
A 100 g granite cube slides down a 40 frictionless ramp. At the bottom, just as it exits onto a horizontal table, it collides with a 200 g steel cube at rest. How high above the table should the...
-
You have been asked to design a ballistic spring system to measure the speed of bullets. A spring whose spring constant is k is suspended from the ceiling. A block of mass M hangs from the spring. A...
-
In FIGURE P11.57, a block of mass m slides along a frictionless track with speed v m . It collides with a stationary block of mass M. Find an expression for the minimum value of v m that will allow...
-
Your company is scheduled to receive 2,000,000 euros in 1 year. The euro is currently trading at 1.0813, you can borrow in the US for 1 year at 5.5% or invest in the US at 5.2%. You can borrow in...
-
The APR on a 14-day loan from Moneytree is: Group of answer choices .05% Okay. Give me a hint. What's an APR? 17% 460%
-
Risk management strategies in stock exchange in canada presentation with good points of this topic

Study smarter with the SolutionInn App


