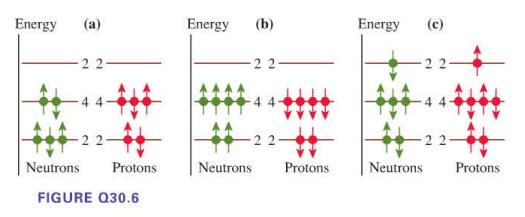
For each nuclear energy-level diagram in Figure Q30.6, state whether it represents a nuclear ground state, an
Question:
For each nuclear energy-level diagram in Figure Q30.6, state whether it represents a nuclear ground state, an excited nuclear state, or an impossible nucleus.
Transcribed Image Text:
Energy (a) 22 + 22- Neutrons Protons FIGURE Q30.6 Energy (b) -22 Neutrons Protons Energy Neutrons (c) 44- 22- Protons
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
a This diagram represents an excited nuclear state The ...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

College Physics A Strategic Approach
ISBN: 9780321595492
2nd Edition
Authors: Randall D. Knight, Brian Jones, Stuart Field
Question Posted:
Students also viewed these Physics questions
-
Seven possible transitions are identified on the energy level diagram in Figure Q29.20. For each, is this an allowed transition? If allowed, is it an emission or an absorption transition, and is the...
-
The CN molecule has been found in interstellar space. Assuming the electronic structure of the molecule can be described using the molecular orbital energy level diagram in Figure 9.16, answer the...
-
In a nuclear physics experiment, a proton is fired toward a Z = 13 nucleus with the diameter and neutron energy levels shown in Figure 40.17 . The nucleus, which was initially in its ground state,...
-
Differentiate between formal and behavioral roles, and describe how behavioral roles emerge during group interaction.
-
Merchandise is sold on account to a customer for $18,000, terms FOB shipping point, 3/10, n/30. The seller paid the transportation costs of $375. Determine the following: (a) Amount of the sale, (b)...
-
Refer to Problem 1. For each of the five payment schedules, determine the present worth of the loan payments made by the borrower. Use an Excel spreadsheet and program it such that you can enter...
-
Increasingly, we are seeing email used in cases involving defendants located in foreign countries. Plaintiffs filed suit against four Defendants: Qingdao Sunflare New Energy Co., Skone Lighting Co.,...
-
Southworth Company uses a job-order costing system and applies manufacturing overhead cost to jobs on the basis of the cost of direct materials used in production. At the beginning of the current...
-
You are the owner of Poptastic. For the past few years, to help supplement your familys income, you have sold premium, homemade popsicles from your food truck and a few local businesses in Pensacola,...
-
A sample contains a mix of isotopes of an element. Using a spectrometer to measure the spectrum of emitted light will not reveal the mix of isotopes; analyzing the sample with a mass spectrometer...
-
A radioactive sample has a half-life of 10 s. 10,000 nuclei are present at t = 20 s. a. How many nuclei were there at t = 0 s? b. How many nuclei will there be at t = 40 s?
-
Imagine a collision between an oxygen, O 2 , molecule and a hydrogen, H 2 , molecule to give the product hydrogen peroxide, H 2 O 2 . Draw Lewis dot diagrams for all the molecules, and then indicate...
-
Padgett invests in Bryant Inc., a glass manufacturing company. As a stockholder, he owns a part of the company and he holds the right to vote on company issues. However, he is entitled to dividends...
-
Feeling hungry, you decide to take a deep dive into data from your local town regarding estimated tons of oranges for each radius mile away from you (from 1 to 5). Optimistically, you believe that...
-
how to complete the Statement of Cash Flows ACC 122 Spring 2021 Comprehensive Project BestValue Corporation's Trial Balance at December 31, 20XX is presented below. All 20XX transactions have been...
-
d . The metal with the highest energy Fermi level from the options below is: i . Pb , Valency ( q ) = 4 , molar volume 1 8 . 2 6 cm - 3 ii . Al , Valency ( q ) = 3 , molar volume 1 0 cm - 3 iii. Zn ,...
-
I. Explain Requirements Engineering Process. I [04] [10 Marks] II. Explain System Modeling, its benefits and explain various types of modeling. [05] [10 Marks] III. Explain Prototyping and its role...
-
The following events were reported in the financial statements of large, publicly owned corporations: a. Atlantic Richfield Company (ARCO), previously a separate company that is now owned by BP...
-
During the month, services performed for customers on account amounted to $7,500 and collections from customers in payment of their accounts totaled $6,000. At the end of the month, the Accounts...
-
A wave travels with speed 200 m/s. Its wave number is 1.5 rad/m. What are its (a) Wavelength (b) Frequency?
-
You are standing at x = 0 m, listening to a sound that is emitted at frequency f 0 . The graph of FIGURE Q16.12 shows the frequency you hear during a 4-second interval. Which of the following...
-
The displacement of a wave traveling in the negative y-direction is D(y, t) = (5.2 cm) sin(5.5y + 72t), where y is in m and t is in s. What are the (a) Frequency, (b) Wavelength, (c) Speed of this...
-
What is the role of cost accounting in managerial decision-making processes?
-
Discuss the impact of GAAP (Generally Accepted Accounting Principles) on financial reporting standards.
-
Provided $17,300 in animal care services for customers on April 3, all on credit. What accounts would this effect?

Study smarter with the SolutionInn App


