Seven possible transitions are identified on the energy level diagram in Figure Q29.20. For each, is this
Question:
Seven possible transitions are identified on the energy level diagram in Figure Q29.20. For each, is this an allowed transition? If allowed, is it an emission or an absorption transition, and is the photon infrared, visible, or ultraviolet? If not allowed, why not?
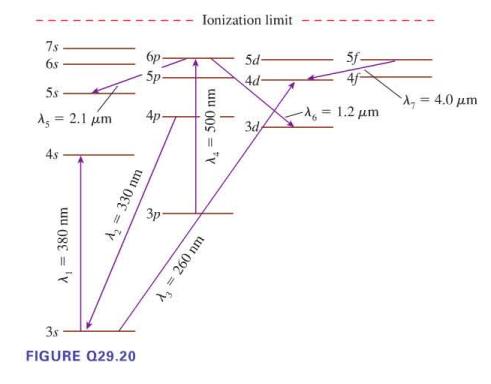
Transcribed Image Text:
7s 68 5s A = 2.1 μm 4s A₁ = 380 nm A₂ = 330 nm 3.s FIGURE Q29.20 бр 5p- 3p Ionization limit A₂ = 500 nm A₂ = 260 nm 5d- 4d- 3d/ 5f- -λ = 1.2 μm = 4.0 μm
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
Here is an analysis of the seven possible transitions identified on the energy level diagram in Figure Q2920 Transition Allowed Type of Transition Typ...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

College Physics A Strategic Approach
ISBN: 9780321595492
2nd Edition
Authors: Randall D. Knight, Brian Jones, Stuart Field
Question Posted:
Students also viewed these Physics questions
-
Figure 28.19 shows part of the energy level diagram for the electrons in an imaginary atom. The arrows represent three transitions between the energy levels. For each of these transitions: a....
-
Question 1 [34 marks] Read the scenarios below for the year ending 28 February 2023 and answer the following question. A. The following is the income statement of Axe Ltd for the financial year...
-
In a set of experiments on a hypothetical one-electron atom, you measure the wavelengths of the photons emitted from transitions ending in the ground state (n = I), as shown in the energy level...
-
Were Napoleon's territorial ambitions significantly different from pre-1799 conquests? If so, where?What were Napoleon's most significant domestic accomplishments in France? Consider the interesting...
-
Zippy Computers announced strong fourth quarter results. Sales and earnings were both above analysts expectations. You notice in the newspaper that Zippys stock price went up sharply on the day of...
-
The cylinder in Fig. P2.154 accelerates to the left at the rate of \(9.80 \mathrm{~m} / \mathrm{s}^{2}\). Find the tension in the string connecting at rod of circular cross section to the cylinder....
-
The following information was obtained in an audit of the cash account of Tuck Company as of December 31, 19X0. Assume that the CPA has satisfied himself as to the validity of the cash book, the bank...
-
On January 1, Year 5, Pic Company acquired 7,500 ordinary shares of Sic Company for $600,000. On January 1, Year 6, Pic Company acquired an additional 2,000 ordinary shares of Sic Company for...
-
Bonita Inc. had a bad year in 2019. For the first time in its history, it operated at a loss. The company's income statement showed the following results from selling 76,000 units of product: net...
-
List the quantum numbers of (a) All possible 3p states (b) All possible 3d states.
-
Show, by actual calculation, that the Bohr radius is 0.0529 nm and that the ground-state energy of hydrogen is -13.60 eV.
-
Steel balls \(10 \mathrm{~mm}\) in diameter are annealed by heating to \(1150 \mathrm{~K}\) and then slowly cooling to \(450 \mathrm{~K}\) in an air environment for which \(T_{\infty}=325...
-
3. Suppose that (Xn)nzo is a Markov chain on a state space I = {1, 2, 3, 4, 5}, initial distribution A = = (1/3, 0, 0, 1/6, 1/2) and stochastic matrix [1/4 1/4 1/4 0 1/4] 0 0 5/6 0 1/6 P = 1/3 0 1/5...
-
The "Best Independent Set" problem is defined as follows: Input: An undirected graph G, in which each vertex is marked by a positive value; and a target value T. Goal: A set of vertices S such that...
-
Agri-Panel Inc. is a manufacturer of high-density fibreboard panels. The company, based in Manitoba, has been in business for nearly 28 years and is owned equally by two brothers, Derek and Alex...
-
Assume the robot's initial pose is [xy 0]=[0 0 the trajectory below autonomously. > 20 15 10 5 in 0 -10 -15 -20 20 30 X 2 then robot will move along 50 60 27 The equation for the above trajectory is...
-
For the system shown in Fig 1, M(f) MA 33 16:3.34 Y(D) Fig 1 BPF cos(2m10*t) 5(0) a- What is the bandwidth of m(t)? b- Sketch Y(f). c-Sketch S(f) if the ideal BPF filter has center frequency of 12.5k...
-
RAF Inc. reported 2016 earnings per share of $3.20 and had no discontinued operations items. In 2017, EPS on income from continuing operations was $2.99, and EPS on net income was $3.49. Is this a...
-
Use critical values to test the null hypothesis H0: 1 2 = 20 versus the alternative hypothesis H0: 1 2 20 by setting a equal to .10, .05, .01, and .001. How much evidence is there that the...
-
A rocket in deep space has an exhaust-gas speed of 2000 m/s. When the rocket is fully loaded, the mass of the fuel is five times the mass of the empty rocket. What is the rockets speed when half the...
-
A tennis player swings her 1000 g racket with a speed of 10 m/s. She hits a 60 g tennis ball that was approaching her at a speed of 20 m/s. The ball rebounds at 40 m/s. a. How fast is her racket...
-
A 60 g tennis ball with an initial speed of 32 m/s hits a wall and rebounds with the same speed. FIGURE P11.39 shows the force of the wall on the ball during the collision. What is the value of F max...
-
Set up costs 30,000 Order receiving costs 1,195,00 O Packing 250,000 Engineering 373,000 Total overheads 1,848,00 O The direct costs per unit for products X, Y and Z are; X Y Z Direct Labour (E) 8 12...
-
Does the permission of a resources group is inherited to all the resources in that resource group? Explain and provide reasons.
-
2. The City of Odessa maintains its books so as to prepare fund accounting statements and prepares worksheet adjustments in order to prepare government-wide statements. As such, the City's internal...

Study smarter with the SolutionInn App


