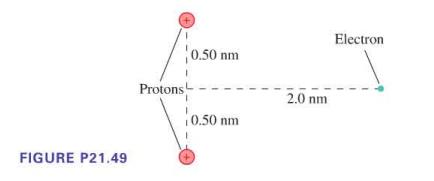
What is the electric potential energy of the electron in Figure P21.49? The protons are fixed and
Question:
What is the electric potential energy of the electron in Figure P21.49? The protons are fixed and can't move.
Transcribed Image Text:
FIGURE P21.49 Protons 0.50 nm 0.50 nm 2.0 nm Electron
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
To determine the electric potential energy of the electron in Figure P2149 consider the following st...View the full answer

Answered By

Saud Ur Rehman
Evaluating manufacturing processes by designing and conducting research programs; applying knowledge of product design, fabrication, assembly, tooling, and materials; conferring with equipment vendors; soliciting observations from operators. Developing manufacturing processes by studying product requirements; researching, designing, modifying, and testing manufacturing methods and equipment; conferring with equipment vendors. Keeping equipment operational by coordinating maintenance and repair services; following manufacturer's instructions and established procedures; requesting special service.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

College Physics A Strategic Approach
ISBN: 9780321595492
2nd Edition
Authors: Randall D. Knight, Brian Jones, Stuart Field
Question Posted:
Students also viewed these Physics questions
-
What is the electric potential energy of the electron in the figure? The protons are fixed and cannot move. Assume that r = 0.60nm ?and d = 2.0nm . (Figure 1)
-
Answer the following questions; 1. What is the difference between a scientific law and a scientific theory? 2. How do scientists arrive at these conclusions, and what are the limitations of...
-
An atom of Na can combine with an atom of Cl to form the molecule NaCl. It is a good approximation to view this process as two steps (Fig. P18.87) in which the Na atom gives up an electron to the Cl...
-
?Claim: A of adults would erase all of their personalinformation online if they could. A software firm survey of 511randomly selected adults showed that 36 ?% of them would erase allof their pe...
-
Referring to Table 13.2, calculate the market value of firm L (without a corporate income tax) if the equity amount in its capital structure decreases to $5,000 and the debt amount increases to...
-
Essence of Skunk Fragrances, Ltd., sells 6,700 units of its perfume collection each year at a price per unit of $215. All sales are on credit with terms of 1/10, net 30. The discount is taken by 60...
-
The feed to a distillation tower consists of \(14.3 \mathrm{kmol} / \mathrm{hr}\) of methanol, \(105.3 \mathrm{kmol} / \mathrm{hr}\) of toluene, \(136.2 \mathrm{kmol} / \mathrm{hr}\) of ethylbenzene,...
-
Johnson, Inc., is a manufacturer of lead crystal glasses. The standard materials quantity is 0.8 pound per glass at a price of $0.30 per pound. The actual results for the production of 6,900 glasses...
-
Answer the following: 1. Open the following two video clips link below and write 2 personal reflections on them . Minimum of 6 sentences PER reflection References: Video Clip of a Fixed Action...
-
A pair of 10 F capacitors in a high-power laser are charged to 1.7 kV. a. What charge is stored in each capacitor? b. How much energy is stored in each capacitor?
-
A capacitor with its plates separated by paper stores 4.4 nC of charge when it is connected to a particular battery. An otherwise identical capacitor, but with its plates separated by Pyrex glass, is...
-
Maria meets all of the requirements of 1237 (subdivided realty). In 2015, she begins selling lots and sells four separate lots to four different purchasers. She also sells two contiguous lots to...
-
Sandra would like to organize LAB (a legal corporation) as either an S corporation or a C corporation for tax purposes. In either form, the entity is expected to generate an 8 percent annual...
-
At January 1, Y3, Mapleton Corp.'s defined benefit pension plan, under IFRS, had a defined benefit obligation of $ 100,000, while the fair value of the plan assets was $ 120,000. During Y3, the...
-
On January 1, 2023, Bertrand, Incorporated, paid $90,400 for a 40 percent interest in Chestnut Corporation's common stock. This investee had assets with a book value of $248,500 and liabilities of...
-
Assume that the cost per unit to ship is $9.00 for Origin of Cleveland to all four destinations of Boston, Chicago, St. Louis, and Lexington. What is the minimal cost of transportation if the cost...
-
What is Tim's refund? A dependent can claim as a deduction the higher of the two amounts: $850, or the amount of earned income plus $300, up to $5,450. Tim is a dependent who earned $410 part-time...
-
Repeat for the 20082009 NFL seasons. In exercise, the home and away attendance figures for the National Football League for the 20122013 seasons were recorded. a. Analyze the relationship between the...
-
Tiger, Inc. signed a lease for equipment on July 1, 2007.The lease is for 10 years (the useful life of the asset).The first of 10 equal annual payments of $500,000 was made on July 1, 2007.The...
-
Consider the compost bed shown in the right-hand figure. The dimensions of the bed are 1 m by 2 m. A 10-cm (0.10-m) thick layer of organic compost lines the bottom as shown. Slow bacterial...
-
Consider a section of muscle tissue of cylindrical shape with a radius of 1.5 cm. During highly rigorous exercise, metabolic processes generate 15 kW/m 3 of bulk tissue. The outer surface of the...
-
A 1-in.-OD steel tube has its outside wall surface maintained at 250F. It is proposed to increase the rate of heat transfer by adding fins of 3/32-in. thickness and 3/4-in. long to the outside tube...
-
How does metatheoretical reflexivity enhance the self-awareness and adaptability of critical approaches, and what challenges does it pose?
-
Profit equals: O (P x Q-V x Q) + fixed expenses. O (P x Q-V x Q) - fixed expenses. O (P-V-fixed expenses) Q. O (P x Q + V x Q) - fixed expenses
-
A pile of cards contains eight cards, numbered 1 through 8. What is the probability of NOT choosing the 6? 118 - 100 0|1 O Mark this question -1/13 UN 5.1

Study smarter with the SolutionInn App


