Calculate the energy released in the fission reaction of Eq. (29-47). on + 235U 235U* 230U*
Question:
Calculate the energy released in the fission reaction of Eq. (29-47).
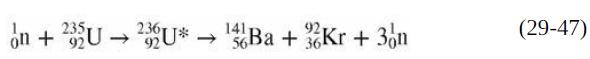
Transcribed Image Text:
on + 235U → 235U* 230U* 14Ba + Kr + 3n (29-47)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
mass of reactant 1235 236 mass of p...View the full answer

Answered By

Brian Otieno
I'm Brian , an experienced professional freelancer with countless hours of success in freelancing many subjects in different disciplines. Specifically, I have handled many subjects and excelled in many disciplines. I have worked on many Computer Science projects and have been able to achieve a lot in that field. Additionally, I have handled other disciplines like History, Humanities, Social Sciences, Political science, Health care and life science, and Religion / Theology. My experience generally in these subjects has made me able to deliver high-quality projects in a very timely fashion. I am very reliable at my job and will get the work done in time, no matter what. In Addition, I have managed to ensure that the work meets my client's expectations and does not cause an error. I am a hard-working and diligent person who is highly responsible for everything I do. Generally, Freelancing has made me more accountable for doing my job. Additionally, I have had a passion for writing for the last seven years in this field.
0.00
0 Reviews
10+ Question Solved
Related Book For 

College Physics With An Integrated Approach To Forces And Kinematics
ISBN: 978-1260547719
5th Edition
Authors: Alan Giambattista
Question Posted:
Students also viewed these Sciences questions
-
Calculate the energy released in the fission reaction of Eq. (29-30). The atomic masses of 14156Ba and 9236Kr are 140.914 u and 91.926 u, respectively.
-
Calculate the energy released in the fission reaction 235U + n ( 141Cs + 93Rb + 2n. Here are some atomic and particle masses. 2235.043 92 u 92.921 57 u 14Cs 140.919 63 n 1.008 66 u
-
Calculate the energy released in the fission reaction n + 235/92U 88/38Sr + 136/54Xe + 12n. Use Appendix B, and assume the initial kinetic energy of the neutron is very small.
-
trade on the common stock of Taz, Inc. that have a strike price of $ 51.00 and a premium of $ 1.00 . In each of the next four parts, calculate the net profit (or loss) on the option position. Note:...
-
A total of 34,695 adults were asked whether they agreed with the following statement: I often feel that my values are threatened by Hollywood and the entertainment industry. Table 18 compares the...
-
Ming Company uses a perpetual inventory system. It entered into the following purchases and sales transactions for April. (For specific identification, the April 9 sale consisted of 8 units from...
-
An immersion heater operating at \(1000 \mathrm{~W}\) is in the form of a rectangular solid with dimensions of \(16 \mathrm{~cm}\) by \(10 \mathrm{~cm}\) by \(1 \mathrm{~cm}\). Determine the heat...
-
J. K. Pratt Co. had the following transactions: 20-1 July 20 Received a $750, 30-day, 10% note from J. Akita in payment for sale of merchandise. Aug. 19 J. Akita paid note issued July 20 plus...
-
Explain how tax-benefit linkages, spillovers, and economies of scale influence fiscal federalism (that is, what each of those implies in terms of the optimal division of responsibilities across the...
-
An aircraft has to fly between two cities, one of which is 600.0 km north of the other. The pilot starts from the southern city and encounters a steady 100.0 km/h wind that blows from the northeast....
-
One possible fission reaction for 235 U is 235 U + n 141 Cs + 93 Rb + ?n, where ?n represents one or more neutrons. (a) How many neutrons? (b) From the graph in Fig. 29.2, you can read the 7 5...
-
Estimate the energy released in the fission reaction of Eq. (29-48) from the values of the binding energy per nucleon in Fig. 29.2. 8 7 5 Binding energy per nucleon (MeV) 3 + go 2C He Li He 2 1 H Ni...
-
How do the human resource practices at UPS help build teamwork
-
At the beginning of September Barney & Co were owed 200 in rent. At the end of September, they were owed 400. 800 cash for rent was received during September. What entry will be made in the statement...
-
Jackson County Senior Services is a nonprofit organization devoted to providing essential services to seniors who live in their own homes within the Jackson County area. Three services are provided...
-
(10) A person earns a wage $10 per hour and has 100 hours per week to allocate between leisure and work. At the $10 wage, the person chooses to consume 65 hours of leisure per week. a. Draw the...
-
With a downsloping demand curve and an upsloping supply curve for a product, an increase in consumer income will: a . decrease equilibrium price and quantity if the product is a normal good. b . have...
-
3. A crate of mass 45kg was pushed from rest by a horizontal force of 120N on a horizontal plane from point A to point B. The distance between A and B was 80m. The friction coefficient between the...
-
Compare a rotary transfer machine with an unmanned robotic cell.
-
A certain Christmas tree ornament is a silver sphere having a diameter of 8.50 cm. Determine an object location for which the size of the reflected image is three-fourths the size of the object. Use...
-
Identify the reagents necessary to achieve each of the following transformations: (a) (b) (c) (d) (e) (f) HO H.
-
Draw a mechanism for each of the following reactions: (a) (b) 1) EtMgBr 2) - I + HCI
-
Draw a mechanism for each of the following reactions: (a) (b) HO 1) EtMgBr 2) H20
-
Name the financial statement where each of the following will appear: (IS) Income Statement; (BS) Balance Sheet; (SCF) Statement of Cash Flows; (N) None. a. Book value of equipment purchased five...
-
Britton Company sold merchandise on account to K. O'Neil for $1,700, plus $80 sales tax. How would Britton Company record this transaction in the sales journal?
-
Tests of controls are designed to determine all of the following, except: Whether or not the control was overridden. How a control was applied? By what means the control was applied? How consistent...

Study smarter with the SolutionInn App


