Of the hypothetical nuclear reactions listed here, which would violate conservation of charge? (a) (b) Na +
Question:
Of the hypothetical nuclear reactions listed here, which would violate conservation of charge?
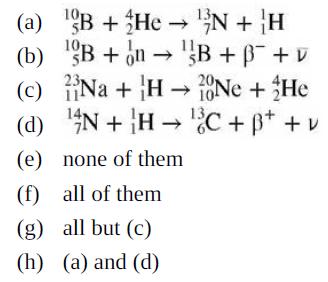
Transcribed Image Text:
(a) (b) Na + H → (c) (d) ¹N+H → (e) none of them (f) all of them (g) all but (c) (h) (a) and (d) ¹B+He B+ on → N + H ¹B + ߯ + v 20 -10¹ 13, Ne + He C + B+ + v
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 37% (8 reviews)
In all cases except c the sum of a...View the full answer

Answered By

Marvine Ekina
Marvine Ekina
Dedicated and experienced Academic Tutor with a proven track record for helping students to improve their academic performance. Adept at evaluating students and creating learning plans based on their strengths and weaknesses. Bringing forth a devotion to education and helping others to achieve their academic and life goals.
PERSONAL INFORMATION
Address: , ,
Nationality:
Driving License:
Hobbies: reading
SKILLS
????? Problem Solving Skills
????? Predictive Modeling
????? Customer Service Skills
????? Creative Problem Solving Skills
????? Strong Analytical Skills
????? Project Management Skills
????? Multitasking Skills
????? Leadership Skills
????? Curriculum Development
????? Excellent Communication Skills
????? SAT Prep
????? Knowledge of Educational Philosophies
????? Informal and Formal Assessments
0.00
0 Reviews
10+ Question Solved
Related Book For 

College Physics With An Integrated Approach To Forces And Kinematics
ISBN: 978-1260547719
5th Edition
Authors: Alan Giambattista
Question Posted:
Students also viewed these Sciences questions
-
Of the hypothetical nuclear reactions listed in Multiple-Choice Question 4, which would violate conservation of nucleon number? Data From Question 4 (a) B+He N + H B+nB (b) + + v Na + H 20 10 Ne +...
-
The following equations are for nuclear reactions that are known to occur in the explosion of an atomic bomb. Identify X. 140 235 144
-
Which reagent in each pair listed here would be the more reactive nucleophile in a polar aprotic solvent? (a) CH3H or CH3NH2 (b) CH3O- or CH3CO2- (-OAc) (c) CH3SH or CH3OH (d) (C6H5)3N or (C6H5)3P...
-
Angela Monterey has a five-year car loan for a Jeep Wrangler at an annual interest rate of 6.7% and a monthly payment of $609.50. After 3 years, Angela decides to buy a new car. What is Angela's loan...
-
In 1994, California passed a three-strikes sentencing law man- dating that defendants convicted of a third felony should serve a state prison term of 25 years to life. The density histogram in Fig....
-
A U.S. Treasury bond pays a 7% coupon on January 7 and July 7. How much interest accrues per \($100\) of principal to the bond holder between July 7, 2013, and August 9, 2013? How would your answer...
-
The budgeted monthly service revenues for Shumin Services Ltd for January to June are as follows. All of the firms invoicing for services is on a credit basis. All payments received from clients on...
-
Five kilograms of a 30 wt% acetone70% water mixture is added to 3.5 kg of a 20 wt% ace tone80% MIBK mixture at 25 C. Use Figure 6.6-1 to estimate the mass and composition of each phase of the...
-
The Royal Gorge Bridge in Colorado rises 321 m above the Arkansas River. Suppose you kick a rock horizontally off the bridge. The magnitude of the rock's horizontal displacement is 45.0 m. Find the...
-
Allie has bought a new apple orchard. The orchard has a single file of trees, numbered from 1 to N. Each tree has a certail number of ripe apples. Allie has a rule she wants to follow. She wants to...
-
In a fusion reaction, two deuterons produce a helium-3 nucleus. What is the other product of the reaction? (a) An electron (b) A proton (c) A neutron (d) An alpha particle (e) A positron (f) A...
-
Explain why neutron-activated nuclides tend to decay by rather than + .
-
A coil with an inductance of 2.0 H and a resistance of 10 is suddenly connected to an ideal battery with = 100 V. At 0.10 s after the connection is made, what is the rate at which (a) Energy is...
-
4. A patient admitted to the hospital for surgery tells the nurse, I do not feel comfortable leaving my children with my parents. Which action would the nurse take next? a. Reassure the patient that...
-
7. Which statement describes the purpose of the assessment phase of the nursing process? a. To teach interventions that relieve health problems b. To use patient data to evaluate patient care...
-
Crystal Company produces glass jars. The unit for costing purposes is a case of 1 8 jars. The following standards for producing one case of jars have been established: Direct materials ( 5 kg @ $ 0 ....
-
Vernon Company began operations on January 1 , year 1 , by issuing common stock for $ 3 1 , 0 0 0 cash. During year 1 , Vernon received $ 6 2 , 2 0 0 cash from revenue and incurred costs that...
-
The year - end balance sheet of Social Systems, Inc. includes the following stockholders equity section ( with certain details omitted ) : Stockholders equity: 9 % cumulative preferred stock, Rs . 1...
-
Find a 2 2 matrix E with entries between 0 and 1 such that: I - E has an inverse but not all entries of (I - E)-1 are nonnegative.
-
Read the following description and Write a response of it. The discretion of public administrators can be decreased, but not altogether eliminated. Officials will use their discretion in any given...
-
Consider the structure of beta-carotene, mentioned earlier in this chapter: Design a synthesis of beta carotene using the compound below as your only source of carbon atoms: B-carotene Br
-
Identify the reagents necessary to accomplish each of the transformations below: (a) (b)
-
Predict the major product of each reaction below: (a) (b) (c) RCO3H RCO, ? H RCO3H
-
Who am I communicating to? Meeting 1 (e.g. with Project team) Meeting 2 (e.g. with customers) How does the forum meet organisational objectives? What vocabulary, tone, structure and style suits...
-
A company has issued a bond with a par value of $1,000 and with coupon rate of 6% paid semi-annually with maturity of ten years. A) What is the bond's price after a year if similar risk bonds has...
-
Consider an Investment Universe made of 3 stocks S1, S2 and S3 with the following characteristics: 0.010 0.002 0.001 Covariance matrix: = 0.002 0.011 0.003 0.001 0.003 0.020, P1 4.27% Expected Return...

Study smarter with the SolutionInn App


