What is the gauge pressure of the gas in the closed tube in the figure? (Take the
Question:
What is the gauge pressure of the gas in the closed tube in the figure? (Take the atmospheric pressure to be 760 mmHg.)
(a) 200 mmHg
(b) −200 mmHg
(c) 960 mmHg
(d) 560 mmHg
(e) −960 mmHg −
(f) 560 mmHg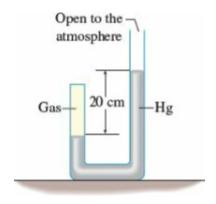
Transcribed Image Text:
Open to the atmosphere -Hg Gas-- 20 cm
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
The gauge pressure of the gas in the closed tube is given by the difference betw...View the full answer

Answered By

BillClinton Muguai
I have been a tutor for the past 5 years. I have experience working with students in a variety of subject areas, including computer science, math, science, English, and history. I have also worked with students of all ages, from elementary school to college. In addition to my tutoring experience, I have a degree in education from a top university. This has given me a strong foundation in child development and learning theories, which I use to inform my tutoring practices.
I am patient and adaptable, and I work to create a positive and supportive learning environment for my students. I believe that all students have the ability to succeed, and it is my job to help them find and develop their strengths. I am confident in my ability to tutor students and help them achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

College Physics With An Integrated Approach To Forces And Kinematics
ISBN: 978-1260547719
5th Edition
Authors: Alan Giambattista
Question Posted:
Students also viewed these Sciences questions
-
The gauge pressure of the air in an automobile tire is 32 lb / in2. Convert this to (a) Pa, (b) Torr, (c) Atm?
-
The gauge pressure of the air in an automobile tire is 32 lb/in 2 . Convert this to (a) Pa, (b) mmHg, (c) atm.
-
Find the minimum attainable pressure of ideal gas in the process T = To + V2, where To and are positive constants, and V is the volume of one mole of gas. Draw the approximate p vs V plot of this...
-
what is the name of Dc output symbol on eagle in this circuit? XL2 EE20 core Flyback Transformer 4 SCHOTTKY [ 3 D4 SB160 IC2 EL817 OPTOCOUPLER thS 1 2 C2 470uF 25V R3 Tko ZD1 12V/1W DC Output 12V,...
-
What monthly payment is required to pay off a $50,000 loan in seven years, if the interest rate on the loan is 7.5% compounded: a. Annually? b. Semiannually? c. Quarterly? d. Monthly?
-
What are the two main types of traditional cost accounting systems?
-
A company manufactures geometrically similar airplane propellers up to \(4.0 \mathrm{~m}\) in diameter. Wind tunnel tests are run on geometrically similar propellers up to \(0.33 \mathrm{~m}\) in...
-
Carol Keene, corporate comptroller for Dumaine Industries, is trying to decide how to present Property, plant, and equipment in the balance sheet. She realizes that the statement of cash flows will...
-
A 5.2 x10 -4 V/m electric field creates a 2.0 x10 17 electrons/s current in a 1.5-mm-diameter aluminum wire. A. What is the drift speed? Express your answer using two significant figures. B. What is...
-
Two wires P and Q both obey Hookes law. They are both stretched and have the same strain. The Young modulus of P is four times larger than that of Q. The diameter of P is twice that of Q. What is the...
-
When helium weather balloons are released, they are purposely underinflated. Why?
-
A manometer contains two different fluids of different densities. Both sides are open to the atmosphere. Which pair(s) of points in the figure have equal pressure? (a) P1 = P5 (b) P2 = P5 (c) P3 = P4...
-
Jer Inc.s $10 par value common stock is actively traded at a market value of $15 per share. Jer issues 5,000 shares to purchase land advertised for sale at $85,000. Journalize the issuance of the...
-
Complete the statements below regarding tax legislation. When enacting tax legislation, Congress is often guided by the concept of revenue neutrality so that any changes the net revenues raised under...
-
Based on the job sheets for the completed production campaigns, how much manufacturing overhead (MOH) was applied to all completed jobs during the year?
-
Go to the web site of your favorite Not-For-Profit organization. Review the site including their mission, services provided, how they raise funding, and their financial reports (if available.) Share...
-
5. Suppose you can earn 10% interest compounded annually. How much would you need to invest today (assume it's January 31, 2018) in order to have $1,000 on January 31, 2026? ANSWER ANSWER F N Suppose...
-
How do Ethics Expectations affect Reporting, Roughly only 37% of companies say their organization provides ongoing performance reporting or key performance indicators on this area. True or False, It...
-
DS 14.3.5 contains the results of a 2' experiment that investigates the preparation of polymers by dispersion polymerization in an organic medium. The response variable is the mean diameter of the...
-
Write a program to move a signed number from smaller register to bigger register. Hint: movzx ax, bl Topic: Data Related Operators and Directives in assembly language
-
A metal rod with a diameter of 2.30 cm and length of 1.10 m has one end immersed in ice at 32.0F and the other end in boiling water at 212F. If the ice melts at a rate of 1.32 g every 175 s, what is...
-
2.0 102 m, (a) What is the thermal resistance if the material is asbestos? (b) What is the thermal resistance if the material is iron? (c) What is the thermal resistance if the material is copper?
-
A child of mass 15 kg climbs to the top of a slide that is 1.7 m above a horizontal run that extends for 0.50 m at the base of the slide. After sliding down, the child comes to rest just before...
-
How would Stephanies investing decisions be affected if she were 35 years old? If she were 50 years old? Amount of Insurance Needed. Considering the information in the previous problem, how much life...
-
Jamie Lee and Ross have been hearing many stories recently about acquaintances who are passing away without leaving a will, which made Jamie Lee and Ross anxious to review their estate plan with an...
-
What are the companys top risks, how severe is their impact and how likely are they to occur?

Study smarter with the SolutionInn App


