The boiling point of 1,4-butanediol is 230C. Would you expect this compound to be soluble or insoluble
Question:
The boiling point of 1,4-butanediol is 230°C. Would you expect this compound to be soluble or insoluble in room-temperature water? Explain.
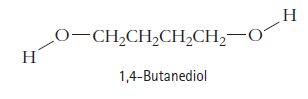
Transcribed Image Text:
Н О-СН,CH,CH,CH, —0 1,4-Butanediol
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
A high boiling point means that the substance inter...View the full answer

Answered By

Arshad Ahmad
Well, I am really new to tutoring but I truly believe a good student can be a better teacher. I have always been a topper at school. I passed my Chartered Accountancy at a very young age of 23, a rare feat for most of the students. I am really dedicated to whatever work I do and I am very strict regarding deadlines. i am always committed and dedicated to whatever work allotted to me and I make sure it is completed well within deadline and also I try to give my best in whatever I do. Hope we will have a good time studying together.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Conceptual Physical Science
ISBN: 978-0134060491
6th edition
Authors: Paul G. Hewitt, John A. Suchocki, Leslie A. Hewitt
Question Posted:
Students also viewed these Physics questions
-
Based on atomic size, which would you expect to be more soluble in water: helium, He, or nitrogen, N 2 ?
-
Why is rain or snow called precipitation?
-
Which of these boxes best represents a suspension?
-
This histogram shows the times, in minutes, required for 25 rats in an animal behavior experiment to successfully navigate a maze. Which of the following best describes the shape of the histogram? A....
-
Pruitt Corporation's overhead costs are usually $20,000 per month. However, the company pays $120,000 of real estate tax on the factory facility in March. Thus, the overhead costs for March Increase...
-
Many organizations are moving to a collaborative process with their major customers to get their input on planning future inventory levels and production rather than relying on an internally...
-
Improve your critical thinking skills.(p. 208)
-
For each of the following situations, two scenarios are described, labeled A and B. Choose which scenario is descriptive of a setting corresponding to activity-based responsibility accounting and...
-
Viking Corporation is owned equally by Sven and his wife Olga, each of whom hold 100 shares in the company, Viking redeemed 80 shares of Sven's stock for $2,200 per share on December 31, 20X3. Viking...
-
The drug Librium gives a polarographic wave with E 1/2 = - 0.265 V (versus S.C.E.) in 0.05 M H 2 SO 4 . A 50.0-mL sample containing Librium gave a wave height of 0.37 A. When 2.00 mL of 3.00 mM...
-
Explain why, for these three substances, the solubility in 20C water goes down as the molecules get larger, but the boiling point goes up. Boiling point/ Solubility Substance 65C CH;-0 infinite 117C...
-
If nitrogen, N 2 , were pumped into your lungs at high pressure, what would happen to its solubility in your blood?
-
What is the net present value of a simple one-period project with an initial investment of $12,000 and an expected net cash flow in one year of $15,000, assuming a discount rate of 8 percent?
-
Question 7 Two objects, of masses 3 and 4 kg, are hung from the ends of a stick that is 70 cm long and has marks every 10 cm, as shown above. If the mass of the stick is negligible, at which of the...
-
Since they do not have enough saved, Rachel and John would like to consider retiring later. Create a new timeline and recalculate all of the relevant values to determine at what age Rachel and John...
-
Problem 6 Find the partial derivative with respect to x for the following functions: (a) p = 56 (b) y(x)=56-4x (c) m = r (d) q= x (e) f(x) =x3 (f) g(x,y) = xy 2 (g) h(x,y) = Ax1/2y1/2, where A is a...
-
Consider the information in the file named Cost Functions of the Firm (also presented above). Please read that file carefully before answering this and the following questions. The fixed cost of...
-
On January 1, 2022, Monica Company acquired 80 percent of Young Company's outstanding common stock for $872,000. The fair value of the noncontrolling interest at the acquisition date was $218,000....
-
Find the remaining trigonometric ratios. cos x = -1/3. < x < 3/2
-
One Way Cellular accountants have assembled the following data for the year ended September 30, 2014: Prepare the operating activities section using the indirect method for One Way Cellulars...
-
Under what conditions does the magnitude of the average velocity equal the average speed?
-
Is it possible for an object to be (a) Slowing down while its acceleration is increasing in magnitude; (b) Speeding up while its acceleration is decreasing? In both cases, explain your reasoning.
-
Canada geese migrate essentially along a northsouth direction for well over a thousand kilometers in some cases, traveling at speeds up to about 100 km/h. If one goose is flying at 100 km/h relative...
-
business law A partner may actively compete with the partnership True False
-
A company provided the following data: Selling price per unit $80 Variable cost per unit $45 Total fixed costs $490,000 How many units must be sold to earn a profit of $122,500?
-
Suppose a 10-year, 10%, semiannual coupon bond with a par value of $1,000 is currently selling for $1,365.20, producing a nominal yield to maturity of 7.5%. However, it can be called after 4 years...

Study smarter with the SolutionInn App


