A stocks returns have the following distribution: Assume the risk-free rate is 2%. Calculate the stocks expected
Question:
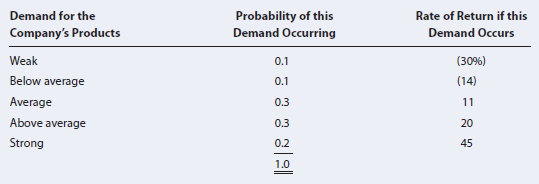 Assume the risk-free rate is 2%. Calculate the stock€™s expected return, standard deviation, coefficient of variation, and Sharpe ratio.
Assume the risk-free rate is 2%. Calculate the stock€™s expected return, standard deviation, coefficient of variation, and Sharpe ratio.
Transcribed Image Text:
Probability of this Demand Occurring Rate of Return if this Demand Occurs Demand for the Company's Products 0.1 0.1 (30%) (14) Weak Below average Average Above average 11 0.3 0.3 20 0.2 45 Strong 1.0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
0130 0114 0311 0320 0245 1390 ...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Fundamentals of Financial Management
ISBN: 978-1337395250
15th edition
Authors: Eugene F. Brigham, Joel F. Houston
Question Posted:
Students also viewed these Business questions
-
A stocks returns have the following distribution: Calculate the stocks expected return, standard deviation, and coefficient of variation. Probability of This Demand Occurring 0.1 0.1 0.3 0.3 Demand...
-
Assume that you have just been hired as business manager of Campus Deli (CD), which is located adjacent to the campus. Sales were $1,100,000 last year, variable costs were 60% of sales, and fixed...
-
Assume that you are considering purchasing stock as an investment. You have narrowed the choice to CDRom.com and Web Stores and have assembled the following data. Selected income statement data for...
-
Listing 8.4 checks whether a solution is valid by checking whether every number is valid in the board. Rewrite the program by checking whether every row, every column, and every small box has the...
-
On January 1, 2014, Morrison Inc., a public company, purchased $600,000 of Pearl Corporation's five year, 4% bonds for $627,660, when the market interest rate was 3%. Interest is received...
-
The following analysis is derived from the research and development (R&D) section of IRON Corporation. A lathe used for the purpose of cutting molded plastics was acquired 10 years ago for a total...
-
Suppose that a forward contract on an asset is written at time zero and there are \(M\) periods until delivery. Suppose that the proportional carrying charge in period \(k\) is \(q S(k)\), where...
-
Distinguish between a client letter of representation and a management letter and state the primary purpose of each. List some items that might be included in each letter.
-
What role does emotional intelligence play in conflict management, and how can emotional regulation techniques enhance conflict resolution effectiveness in interpersonal and organizational contexts ?
-
After Chris completed the ratio analysis for S&S Air (see Chapter 3), Mark and Todd approached him about planning for next years sales. The company had historically used little planning for...
-
Bond X is noncallable and has 20 years to maturity, an 8% annual coupon, and a $1,000 par value. Your required return on Bond X is 9%; if you buy it, you plan to hold it for 5 years. You (and the...
-
a. 1. Why is the T-bills return independent of the state of the economy? Do T-bills promise a completely risk-free return? Explain. 2. Why are High Techs returns expected to move with the economy,...
-
Mallory Corporation has a calendar year-end. The corporation has paid estimated taxes of $10,000 during 2020 but still owes an additional $5,000 for its 2020 tax year. a. When is the 2020 tax return...
-
2. Write balanced half reactions and complete reactions for the following oxidation-reduction reactions. a.* aluminum(s) + hydrogen ion(aq) aluminum ion(aq) + hydrogen (g) oxid. half rxn.: Alis) =...
-
1. Cis- indicates that the functional groups are on the same side. Whereas trans- means that the functional groups are on opposing sides. 2. Cis- and trans- cannot be used in naming alkanes or...
-
A gas mixture consisting of 50 mol% methane and 50 mol% inert gas at 10 atm (1013 kPa) enters a reactor at 422.2 K at a volumetric flow rate of 6 dm3 s"1. Calculate (a) the concentration of methane...
-
How many moles of N2 are produced from exactly 0.1265 moles of Na(N3)2 according to the reaction: 4 Na(N3)2 (s) + O2(g) 12 N2 (g) + 2 Na2O(s)? 0.3795 moles 0.1265 moles O 0.2530 moles 1.518 moles
-
3. The following scheme describes reactions within a fatty acid synthetase. Complete the scheme with correct structures and draw the curved arrows to show how each structure is formed. thlo...
-
Suppose taxpayers are required to pay a base tax of $50 plus 30 percent of any income over $100, as in the initial tax system B in Table 16.1. Suppose further that the taxing authority wishes to...
-
Interview managers at three companies in your area about their use of ERP. How have their experiences been similar? What accounts for the similarities and differences?
-
After a 5-for-1 stock split, Strasburg Company paid a dividend of $0.75 per new share, which represents a 9% increase over last years pre-split dividend. What was last years dividend per share?
-
Northern Pacific Heating and Cooling Inc. has a 6-month backlog of orders for its patented solar heating system. To meet this demand, management plans to expand production capacity by 40% with a $10...
-
Welch Company is considering three independent projects, each of which requires a $5 million investment. The estimated internal rate of return (IRR) and cost of capital for these projects are...
-
This number is the "federal adjusted gross income" according to the line title. What do we call this process of starting our state return with a number from the federal return?
-
What is the main reason for a franchisee to have a contingency fund?
-
Kalan purchased four new tires at Walmart for $1,000 using there "no payments for 12 months plan." About a year later, before Kalan had made any payments, the tires were not performing as well as...

Study smarter with the SolutionInn App


