Using the results of Exercise 4.5, show that = E/ [2(1 + v)] and =
Question:
Using the results of Exercise 4.5, show that μ = E/ [2(1 + v)] and λ = Ev/ [(1 + v) (1 – 2v)].
Data from exercise 4.5
Following the steps outlined in the text, invert the form of Hooke’s law given by (4.2.7) and develop form (4.2.10). Explicitly show that E = μ(3λ + 2μ)/ (λ + μ) and v = λ/ [2(λ + μ)].
Equation 4.2.7
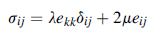
Equation 4.2.10
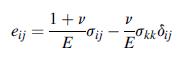
Transcribed Image Text:
dij ;; = ; + 2;; 2Meij
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
From exercise 43 32 2 2 ...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Elasticity Theory Applications And Numerics
ISBN: 9780128159873
4th Edition
Authors: Martin H. Sadd Ph.D.
Question Posted:
Students also viewed these Engineering questions
-
Using the results of Exercise 13.14, verify that the displacement and stress fields for the Boussinesq problem of Example 13.4 are given by (13.4.20) and (13.4.21). Note the interesting behavior of...
-
Using the results of Exercise 11-16, fit the model Y = Bx + E to the chloride concentration-roadway area data in Exercise 11-11. Plot the fitted model on a scatter diagram of the data and comment on...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Google, the immensely popular Web search engine, has been touted as the closest thing the Web has to an ultimate answer machine. Although this is debatable, of course, it is far more difficult to...
-
A string along which waves can travel is 2.70 m long and has a mass of 260 g. The tension in the string is 36.0 N. What must be the frequency of traveling waves of amplitude 7.70 mm for the average...
-
A review of the ledger of Remington Company at December 31, 2010, produces the following data pertaining to the preparation of annual adjusting entries. 1. Salaries Payable $0.There are eight...
-
A structural member in a nuclear reactor is made of a zirconium alloy. If an axial load of 4 kip is to be supported by the member, determine its required cross-sectional area. Use a factor of safety...
-
Analyze IP according to the environmental sustainability portfolio in Figure. What image comes to mind when you hear the words industrial corporation? Pollution-belching smoke stacks? Strip-mined...
-
Oriole Company has the following production data for selected months. Ending Work in Process Beginning Month Work in Process Units Transferred Out % Complete as to Units Conversion Cost January -0-...
-
For isotropic materials show that the principal axes of strain coincide with the principal axes of stress. Further, show that the principal stresses can be expressed in terms of the principal strains...
-
Following the steps outlined in the text, invert the form of Hookes law given by (4.2.7) and develop form (4.2.10). Explicitly show that E = (3 + 2)/( + ) and v = /[2( + )]. Equation 4.2.7 Equation...
-
Write the molecular equation and the net ionic equation for the reaction of calcium carbonate with nitric acid.
-
In Fort Collins, Colorado, the sun shines for an average of 1850 hours per year. A homeowner installs a set of solar panels that provide 4.0 kW of electric power when the sun shines. If the local...
-
Homicide is the killing of a human being by another human being. Most jurisdictions retain the common law definition of the beginning of life: that life begins when a fetus is born alive. There are...
-
If you stuck a paperclip in each of the two slots of a GFI-protected outlet, then grabbed one paperclip with your right hand and the other paperclip with your left hand, would the GFI protect you?...
-
Dana Smith was a notorious drug dealer in a dangerous inner-city neighborhood. One afternoon, Stanley Jacobs visited Danas apartment for the purpose of buying illegal drugs. Dana sold Stanley three...
-
A moving muon lives longer, from the observers point of view, than a muon at rest. Is there a frame of reference in which a muons lifetime is shorter than its lifetime in a frame in which the muon is...
-
Would you rather have a savings account that pays 5% interest compounded semi-annually or one that pays 5% interest compounded Daily? Explain.
-
Multiple Choice Questions: 1. The largest component of aggregate demand is? a. Government purchases. b. Net exports. c. Consumption. d. Investment. 2. A reduction in personal income taxes, other...
-
(a) Write the mass balance for CaCl 2 in water if the species are Ca 2+ and Cl - . (b) Write the mass balance if the species are Ca 2+ , Cl - , CaCl - , and CaOH + . (c) Write the charge balance for...
-
Write the charge and mass balances for dissolving CaF 2 in water if the reactions are CaF,(8) = Ca2+ + 2F Ca?+ + H,O = CAOH+ + H* Ca2+ + F = CaF+ CaF2(s) = CaF2(aq) F + H* = HF(aq) HF(aq) + F = HF,
-
Write charge and mass balances for aqueous Ca 3 (PO 4 ) 2 if the species are Ca 2+ , CaOH + , CaPO - 4 , PO 3- 4 , HPO 2 4 - , H 2 PO - 4 , and H 3 PO 4 .
-
Xenor Corporation introduced a bond in 2002 that offered a coupon rate of 8 1/2%, resulting in coupon payments of $8.50. The bond is scheduled to mature in 2032. If the current going interest rate in...
-
Consider an investor who purchases a Treasury inflation-indexed note with an original principal amount of $100,000, a 6.500 percent annual coupon rate (coupon is paid semiannually), and 10 years to...
-
1. What must be the betas of a portfolio with the following expected returns E (rp) = 18%, 8%, 28%, and 36%, if r = 4% and E(M) 14%? =

Study smarter with the SolutionInn App


