Following the steps outlined in the text, invert the form of Hookes law given by (4.2.7) and
Question:
Following the steps outlined in the text, invert the form of Hooke’s law given by (4.2.7) and develop form (4.2.10). Explicitly show that E = μ(3λ + 2μ)/(λ + μ) and v = λ/[2(λ + μ)].
Equation 4.2.7
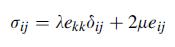
Equation 4.2.10
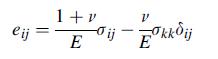
Transcribed Image Text:
gij = kekkoij +2u ij
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
ohe8 2le 322 427 120 e Ou 2 This ...View the full answer

Answered By

John Kimutai
I seek to use my competencies gained through on the job experience and skills learned in training to carry out tasks to the satisfaction of users. I have a keen interest in always delivering excellent work
4.70+
11+ Reviews
24+ Question Solved
Related Book For 

Elasticity Theory Applications And Numerics
ISBN: 9780128159873
4th Edition
Authors: Martin H. Sadd Ph.D.
Question Posted:
Students also viewed these Engineering questions
-
Using the results of Exercise 4.5, show that = E/ [2(1 + v)] and = Ev/ [(1 + v) (1 2v)]. Data from exercise 4.5 Following the steps outlined in the text, invert the form of Hookes law given by...
-
The Hampshire Company manufactures umbrellas that sell for $12.50 each. In 2014, the company made and sold 60,000 umbrellas. The company had fixed manufacturing costs of $216,000. It also had fixed...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
The fraud examiner's report should have what characteristics? a. Accuracy and clarity b. Relevant and material information c. Should not contain opinions or prejudices of fraud examiner. d. All of...
-
A uniform rope of mass m and length L hangs from a ceiling. (a) Show that the speed of a transverse wave on the rope is a function of y, the distance from the lower end, and is given by v = gy. (b)...
-
Givens Graphics Company was organized on January 1, 2010, by Sue Givens. At the end of the first 6 months of operations, the trial balance contained the accounts on the next page. Analysis reveals...
-
Determine the elongation of the square hollow bar when it is subjected to the axial force P = 100 kN. If this force is increased to P = 360 kN and released, find the permanent elongation of the bar....
-
1. What is Johnsons most pressing problem? Why? 2. What can Johnson do to help her team launch the new product within the six-week timeframe? Be specific? 3. Assume Johnson will select a new team to...
-
If the balance of Work in Process on August 31 is $220,000, what was the amount debited to Work in Process for factory overhead in August, assuming a factory overhead rate of 30% of direct labor...
-
For isotropic materials show that the principal axes of strain coincide with the principal axes of stress. Further, show that the principal stresses can be expressed in terms of the principal strains...
-
For isotropic materials, show that the fourth-order elasticity tensor can be expressed in the following forms: Cijkl = ijkl + (ldjk + dik jl) Cjk = ( + ji) + (k u) ijki (oiljk Cijkl E -ijk! + 2(1 +...
-
In testing to find a cure for congenital heart disease, the condition of a patient after he or she has been treated with a drug cannot be directly quantified, but the patient's condition can be...
-
Elizabeth Harris and Paul Thibodeaux lived together with four children. Paul believed he was the biological father of all four children. One afternoon, however, Elizabeth informed Paul that she once...
-
Visit the websites of three Australian companies and access their latest annual reports. View the remuneration report within the directors statutory report. Compare these remuneration reports and...
-
Joey McGrady was driving under the influence of alcohol when he lost control and collided with Melissa Jacobss car. Melissa was 35 weeks pregnant at the time of the accident. After the accident,...
-
The felony murder rule states that a person is guilty of murder when a death results during that persons commission of a felony. Most states limit the felony murder rule to inherently dangerous...
-
A photon striking a solar cell can set one electron in motion in a circuit. For a particular solar cell, the energy delivered to a circuit by each electron is 0.5 eV. Will the energy-conversion...
-
Bill Burne's earnings are subject to deductions for Social Security, Medicare, and FIT. To date his cumulative earnings is $106,700. This week Bill earns $1,200. Bill, who is married, is paid weekly...
-
H Corporation has a bond outstanding. It has a coupon rate of 8 percent and a $1000 par value. The bond has 6 years left to maturity but could be called after three years for $1000 plus a call...
-
Using activities, find the concentrations of the major species in 0.10 M NaClO 4 saturated with Mn(OH) 2 . Take the ionic strength to be 0.10 M and suppose that the ion size of MnOH + is the same as...
-
Explain why the solubility of an ionic compound increases as the ionic strength of the solution increases (at least up to ~ 0.5 M).
-
Which statements are true? In the ionic strength range 00.1 M, activity coefficients decrease with (a) Increasing ionic strength; (b) Increasing ionic charge; (c) Decreasing hydrated radius.
-
Mike's total RRSP contribution room for the current year is $8,000, while his wife Maria's room is $2,000. Mike has decided to contribute $5,000 to his own RRSP and $3,000 to a spousal RRSP for...
-
25 28 30 30 Sheridan Manufacturing Company uses a job order cost system and keeps perpetual inventory records. June 1 Purchased raw materials for $22,800 on account. 8 Raw materials requisitioned by...
-
Imagine a vendor responded to your request for a HRIS proposal. What are the indirect/direct costs and benefits of the HRIS? What is the cost of implementation (include dollar estimates)? Does the...

Study smarter with the SolutionInn App


