A crude model of the water molecule has a negatively charged oxygen atom and two protons, as
Question:
A crude model of the water molecule has a negatively charged oxygen atom and two protons, as shown in Fig. 23.12. Calculate the electrostatic energy of this configuration, which is therefore the magnitude of the energy released in forming this molecule.
Exercise 17
Figure 23.12
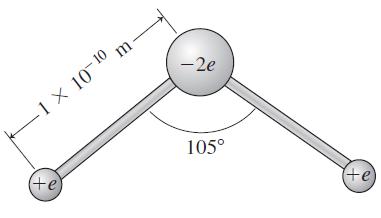
Transcribed Image Text:
-2e -1 × 10¬10 m 105° +e +e FIGURE 23.12 Exercise 17
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (9 reviews)
To determine The electrostatic energy of the water molecule The electrostatic energy of the model of ...View the full answer

Answered By

Kashif Qamar
Learning entails sharing as well. Being a student, I have been sharing my knowledge since high school times because this is what enriches me. I have been on Chegg as a tutor for a long time and also provide lessons to some other students at my localities. Currently, I am also working as a Chemistry and Biology expert at Bartleby.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A model of the water molecule H2O is shown in Figure 9-B. Where is the center of mass? . 52 52
-
Two blocks each of mass m, are attached to the ends of a mass less rod which pivots as shown in Fig.8-40. Initially the rod is held in the horizontal position and then released. Calculate the...
-
Two point charges, Q1 = -6.7μC and Q2 = 1.8μC are located between two oppositely charged parallel plates, as shown in Fig. 16-65. The two charges are separated by a distance of x = 0.34. Assume...
-
1. Was the contract between Joel King and Barber bilateral or unilateral? 2. What was bargained for and given in exchange by each party? Mark P. Painter, Judge This case involves neighbors and the...
-
In Europe, cars' gas consumption is measured in liters per 100 kilometers. In the United States, the unit used is miles per gallon. a) How are these units related? b) How many miles per gallon does...
-
Which of the following is the most accurate statement? a) Like the 20th century, the 21st century will definitely be the American Century. b) Although we have had some recent problems, our economy is...
-
How does make determine whether a target needs to be rebuilt?
-
Fair Value Addison Manufacturing holds a large portfolio of debt and equity securities as an investment. The fair value of the portfolio is greater than its original cost, even though some securities...
-
What are some ways to determine which database management system will work best for a given project? What are some best practices or industry-accepted criteria for matching a given database...
-
Bohrer, CPA (Chartered Professional Accountant), is considering the following factors in assessing audit risk at the financial statement level in planning the audit of Waste Remediation Services...
-
If the three particles in Fig. 23.1 have identical charge q and mass m, and if they?re released from their positions on the triangle, what speed v will they have when they?re far away? ? 92 93 a...
-
A capacitor consists of square conducting plates 25 cm on a side and 5.0 mm apart, carrying charges 1.1 C. Find (a) the electric field, (b) the potential difference between the plates, (c) the stored...
-
What are two reasons why decision-making is required in a programmed work cycle?
-
A Monopoly market consists of One dominating company Two companies Few companies Many similar companies
-
According to the principle of comparative advantage, if a rich country trades with a poor country, then both countries will benefit. O the rich country will lose and the poor country will benefit....
-
The production possibilities frontier is the boundary between O those wants that are limited and those that are unlimited. O those combinations of goods and services that can be produced and those...
-
Consider the amplifier circuit of Fig. 7.15(a) without the load resistance R, and with channel-length modulation neglected. Let Vpp = 5 V, V, = 0.7 V, and k = 1 mA/V. Find Vov. I. R. and R, to obtain...
-
As most plant cells lack lysosomes, what organelle in plant cells performs the functions of recycling and degradation of molecules and old organelles? O chloroplast O central vacuole O starch...
-
Grand Corporation reported pre-tax book income of $600,000. Tax depreciation exceeded book depreciation by $400,000. In addition, the company received $300,000 of tax-exempt municipal bond interest....
-
The following processes constitute the air-standard Diesel cycle: 12: isentropic compression,23: constant-volume energy addition (T and P increase),34: constant-pressure energy addition (v...
-
Amino acids, such as glycine, are the key building blocks of proteins and will be discussed in greater detail in Chapter 25. At the pH of the stomach, glycine exists predominantly in aprotonated form...
-
In each compound below, two protons are clearly identified. Determine which of the two protons is more acidic. (a) (b) (c) (d) N' C-
-
Nitrogen and sulfur are neither in the same row nor in the same column of the periodic table. Nevertheless, you should be able to identify which proton below is more acidic. Explain your choice: .S....
-
A taxpayer disposed a part of asset for 100,000 SR in cash. The taxpayer purchased that asset for 90,000 SR 5 years ago but the market value of the complete assets was as follows: Part disposed...
-
A company maintains debt/equity ratio of 0.6. The flotation cost of equity is 12% and flotation cost of debt it is 6%. The firm is considering a new project which will require $5 million in external...
-
Flight Caf prepares in-flight meals for airlines in its kitchen located next to a local airport. The company's planning budget for July appears below: Flight Caf Planning Budget For the Month Ended...

Study smarter with the SolutionInn App


