The table below lists the wavelengths emitted as electrons in identical square-well potentials drop from various states
Question:
The table below lists the wavelengths emitted as electrons in identical square-well potentials drop from various states n to the ground state. Determine a quantity that, when you plot l against it, should yield a straight line. Make your plot, establish a best-fit line, and use your line to determine the width of the square well.
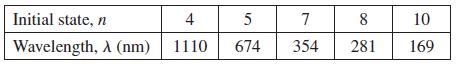
Transcribed Image Text:
Initial state, n 4 5 7 8 10 Wavelength, A (nm) 1110 674 354 281 169
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (12 reviews)
A quantity that when plotted against wavelength should yield a straight line is the r...View the full answer

Answered By

Akshay Shete
I have extensive experience as a tutor, both online and in-person. I have worked with students of all ages and abilities, and am skilled at adapting my teaching style to meet the needs of each individual student. I have a strong background in a variety of subjects, including math, science, and English, and am able to break down complex concepts in a way that is easy for students to understand. In addition to my subject matter expertise, I am also a patient and supportive teacher, and am committed to helping my students succeed. Whether I am working with a struggling student who needs extra help to catch up, or an advanced student looking to get ahead, I am able to provide the guidance and support they need to reach their goals. Overall, my hands-on experience as a tutor has prepared me to be a confident and effective teacher, and I am excited to use my skills to help students succeed.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The table below shows the wavelengths of photons emitted when identical molecules drop from the lth rotational level to the (l - 1)th level. Find quantities that, when plotted, should yield a...
-
The table below lists the stopping potential as a function of wavelength in a photoelectric effect experiment. Determine quantities to plot that should yield a straight line. Make your plot,...
-
Measurements of the potential at points on the axis of a charged disk are given in the two tables below, one for measurements made close to the disk and the other for measurements made far away. In...
-
The Wall Street Journal reported interest rates of 6 percent, 6.35 percent, 6.65 percent, and 6.75 percent for three-year, four-year, five-year, and six-year Treasury notes, respectively. According...
-
List two factors that determine the intensity of an infrared absorption.
-
Assume that you are a salesperson who calls on retailers. For some time, you have been attempting to get an appointment with the head buyer for one of the best retailers in the city to persuade her...
-
Chicago Prime Packers Inc. was a US corporation with its principal place of business in Colorado. Northam Food Trading Company was a Canadian corporation with its principal place of business in...
-
In 2014, Andys Corporation had year-end assets of $2,400,000, sales of $3,300,000, net income of $280,000, net cash flows from operating activities of $390,000, dividends of $120,000, purchases of...
-
a.) The following questions refer to the optical cavity in the diagram below. It is excited by a variable frequency source and the detected intensity is displayed on an Optical Spectrum Analyzer....
-
Smith & Johnson is a supply distributor that used activity-based costing to determine the cost of serving its clients. Different clients demanded different services from Smith & Johnson. For...
-
Youre trying to convince a friend that nuclear energy represents a much more concentrated energy source than fossil fuels, whose combustion involves rearranging atomic electrons. For a rough...
-
The next three problems solve the Schrdinger equation for finite square wells like that shown in Fig. 35.14. Its convenient to work in dimensionless forms of the particle energy E and well depth U0,...
-
The following post-closing trial balance was prepared for Speedy Prints, Inc., but some balances were entered in the wrong column. Prepare a corrected post-closing trial balance. Assume all accounts...
-
Buyer Brad is selling his home to seller Sally with a closing date of January 31. Brad's taxes for the last year were $3,756.75. How much is Sally's share of the taxes, using a 365-day-year statutory...
-
Determine what consolidation entries needed when there are intercompany reciprocal accounts (e.g., accounts payable, accounts receivable) and the impact of these entries on consolidated financial...
-
(5 points) Evaluate the integral and check your answer by differentiating. /[sec sec(x) + cos(x) 5 cos(x) cos(x)] dx = + C
-
Deer and Fishing Company owns $6,000 of office furniture. Accumulated Depreciation had a balance of $3,000 before recording this year's depreciation. Depreciation Expense for the current year is...
-
4. Assume the following for a stock and a call and a put option written on the stock. EXERCISE PRICE = $20 CURRENT STOCK PRICE = $22 VARIANCE = .25 TIME TO EXPIRATION = 4 MONTHS RISK FREE RATE = 3%...
-
Dontae stated that he didn't want to earn any more money because it would "put him in a higher tax bracket." What is wrong with Dontae's reasoning?
-
Write a paper detailing a geographic information system (GIS) of your own design that would utilize data in an original manner.
-
Draw a bond-line structure of the peptide that corresponds with the following sequence of amino acid residues, and identify the N terminus and C terminus: Trp-Val-Ser-Met-Gly-Glu
-
Draw the predominant form of Asp-Lys-Phe at physiological pH.
-
Using three-letter abbreviations, identify all possible acyclic tripeptides containing l-leucine, l methionine, and l-valine.
-
A corporation reported cash or 1 5 , 3 0 0 and total assets of 1 8 0 , 0 0 0 on its balance sheet it's common size present for cash will be?
-
Morty Industries, which uses a process - costing system, adds material at the beginning of production and incurs conversion cost evenly throughout manufacturing. The following information was taken...
-
The following information has been extracted from the financial statements of a company. Net income in 2 0 1 6 = $ 3 , 0 0 0 Annual depreciation in 2 0 1 6 = $ 1 , 0 0 0 Net operating working...

Study smarter with the SolutionInn App


