Question: The differential equation dx/dt = 1/10x(10 -x) - h models a logistic population with harvesting at rate h. Determine (as in Example 6) the dependence
The differential equation dx/dt = 1/10x(10 -x) - h models a logistic population with harvesting at rate h. Determine (as in Example 6) the dependence of the number of critical points on the parameter h, and then construct a bifurcation diagram like Fig. 2.2.12.
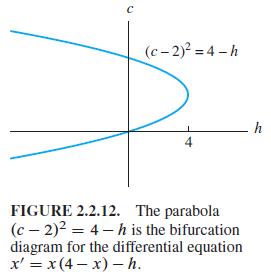
C (c-2) =4-h 4 FIGURE 2.2.12. The parabola (c-2) = 4-h is the bifurcation diagram for the differential equation x' = x(4-x) - h. h
Step by Step Solution
★★★★★
3.44 Rating (154 Votes )
There are 3 Steps involved in it
1 Expert Approved Answer
Step: 1 Unlock

The critical points of the given differential equa... View full answer

Question Has Been Solved by an Expert!
Get step-by-step solutions from verified subject matter experts
Step: 2 Unlock
Step: 3 Unlock


