Find the molar volume of methane at P = 15 bar and T = 200C (it is
Question:
Find the molar volume of methane at P = 15 bar and T = 200°C (it is a gas at these conditions), using the following methods.
A. The Soave equation of state
B. The Peng-Robinson equation of state
C. The virial equation of state
D. The Lee-Kesler generalized correlation
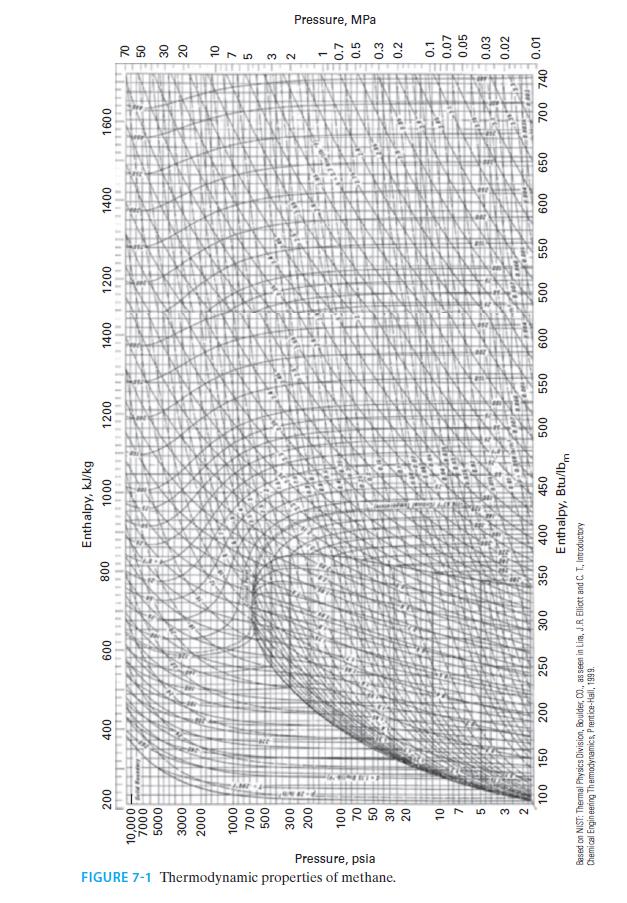
E. Figure 7-1
Transcribed Image Text:
FIGURE 7-1 Thermodynamic properties of methane. Pressure, psia 200 10,000 7000 5000 3000 2000 1000 700 500 300 200 100 70 50 30 20 10 7 5 feld Founday 400 600 800 3 2 100 150 200 250 300 350 Enthalpy, kJ/kg 1000 400 450 Enthalpy, Btu/lbm Based on NIST: Thermal Physics Division, Boulder, 00., as seen in Lima, J.R. Elliott and C. T.. Introductory Chemical Engineering Thermodynamics, Prentice-Hall, 1999. 1200 500 1400 1200 111 550 600 500 1400 550 600 650 1600 700 740 70 50 30 20 27532 10 1 0.7 0.5 0.3 0.2 0.1 0.07 0.05 0.03 0.02 0.01 Pressure, MPa
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
A The Soave equation of state The Soave equation of state is a cubic equation of state that is more accurate than the ideal gas law at high pressures ...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Chemical Engineering Thermodynamics
ISBN: 9781111580704
1st Edition
Authors: Kevin D. Dahm, Donald P. Visco
Question Posted:
Students also viewed these Engineering questions
-
Find the molar volume of nitrogen at P = 10 bar and T = 330 K, using the following methods. A. The Soave equation of state B. The Peng-Robinson equation of state C. The virial equation of state D....
-
Find the molar volume of Freon 22 at P = 5 bar and T = 20C (it is a vapor at these conditions), using the following methods. A. The Soave equation of state B. The Peng-Robinson equation of state C....
-
Which of the following is not a strategic disadvantage of vertical integration? Vertical integration poses all kinds of capacity-matching problems (achieving the most efficient scale of operation for...
-
Refer to Exhibit 10-7 which presents the methodology for analyzing exchange rate variances. Describe in your own words what this methodology accomplishes.
-
Is it necessary to have a written agreement in order to create a partnership?
-
What do you know about the mathematical value of a project's internal rate of return under each of the following conditions? a. The present worth of the project is greater than 0 . b. The present...
-
The income statement of Rodriquez Company is shown below. Additional information: 1. Accounts receivable decreased $310,000 during the year. 2. Prepaid expenses increased $170,000 during the year. 3....
-
quick question are they same " the function of database management system and the characteristics of database management system? explain maintaining data integrity and give simple explanation to...
-
Use the Joback method to estimate T c and P c of each of the following compounds. A. Butane B. 1-Hexanol C. 2-chloropentane D. 3-Hexene E. 1,3-butadiene
-
Using data in Appendix C-1, determine the Peng-Robinson parameters a and b for each of the following compounds at the temperature T = 100C. A. Ethane B. Acetone C. Benzene D. Toluene E. Decane Name...
-
Government survey takers determine that typical family expenditures each month in the year designated as the base year are as follows: 20 pizzas at $10 each Rent of apartment, $600 per month Gasoline...
-
If the list of outcomes includes every possible outcome, then the events are said to be a. independent. b. collectively exhaustive. c. mutually exclusive. d. continuous.
-
Suggest some potential actions that a quantitative analyst could undertake to ensure that the implementation stage of a project is successful.
-
Correlation coefficient measures the a. degree or strength of the linear relationship. b. variability in Y that is explained by the regression equation. c. both a and b. d. none of the above.
-
The values in a probabilistic model a. will always be known with complete certainty. b. can only be determined by developing a new mathematical model. c. will include those that involve an element of...
-
What are the two general forms of time-series models in statistics?
-
Compare Trout Inc. with Salmon Enterprises using the balance sheet of Trout and the market data of Salmon for the weights in the weighted average cost of capital. Salmon Enterprises Bonds Outstanding...
-
a. Why does the Wi-Fi Alliance release compatibility testing profiles in waves instead of combining the entire standards features initially? 27a1.) An 802.11ac Wi-Fi compatibility testing profile...
-
The following data are the measured temperature T of water owing from a hot water faucet after it is turned on at time t = 0. a. Plot the data, connecting them rst with straight lines and then with a...
-
a. Solve the following matrix equation for the matrix C. A(BC + A) = B b. Evaluate the solution obtained in part a for the case 7 9 4 -3 A = -2 4. B = 7 6 ]
-
Solve the following problems using matrix inversion. Check your solutions by computing A -1 A. a. b. c. d. 2 + %3D 5 = 5 9 3 7
-
complete the balance sheet ASSETS = LIABILITIES & OWNERS' EQUITY Cash Filters Vehicles Prepaid Costs Debt Paid-in Capital Retained Earnings YEAR 3 Opening Balances $2,724,276 $472,334 $406,000...
-
There are several reasons why a company would acquire a subsidiary's voting common stock rather than its net assets. Identify at least two advantages to acquiring a controlling interest in the voting...
-
Martinez Company's relevant range of production is 7,500 units to 12,500 units. When it produces and sells 10,000 units, its average costs per unit are as follows: Average Cost per Unit Direct...

Study smarter with the SolutionInn App


