The critical properties for compounds A and B, which are two compounds with similar molecular structures, are
Question:
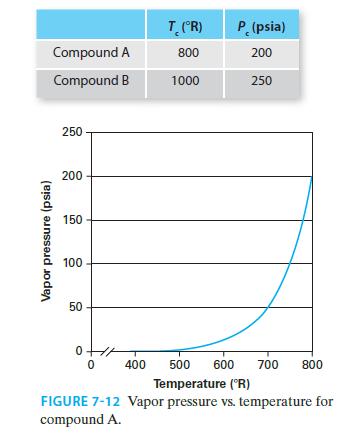
The critical properties for compounds A and B, which are two compounds with similar molecular structures, are given here. No further information is available about compound B, but the vapor pressure of compound A as a function of temperature is given in Figure 7-12. Give the best possible estimates of the vapor pressures for compound B at T = 600°R, 7008R, 800°R, and 900°R.
Transcribed Image Text:
Vapor pressure (psia) Compound A Compound B 250 200 150 100 50 T (R) 800 1000 P. (psia) 200 250 0 0 400 500 600 700 800 Temperature (°R) FIGURE 7-12 Vapor pressure vs. temperature for compound A.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
Step 1 Explore the given information We are asked to estimate vapor pressures for compound B The only direct information we have about compound B is t...View the full answer

Answered By

WAHIDUL HAQUE
hello,
I'm a professional academic solution provider working as a freelance academic solution provider since 7 years. I have completed numerous projects. Help lots of students to get good marks in their exams and quizzes. I can provide any type of academic help to your homework, classwork etc, if you are a student of Accounting, Finance, Economics, Statistics. I believe in satisfying client by my work quality, rather than making one-time profit. I charge reasonable so that we make good long term relationship. why will you choose me? i am an extremely passionate, boldly honest, ethically driven and pro-active contractor that holds each of my clients in high regards throughout all my business relations. in addition, I'll always make sure that I'm giving my 100% better in every work that will be entrusted to me to be able to produce an outcome that will meet my client's standards. so if you are a student that is now reading my profile and considering me for your academic help. please feel free to look through my working history, feedback and contact me if you see or read something that interests you. I appreciate your time and consideration.
regards
4.90+
233+ Reviews
368+ Question Solved
Related Book For 

Fundamentals Of Chemical Engineering Thermodynamics
ISBN: 9781111580704
1st Edition
Authors: Kevin D. Dahm, Donald P. Visco
Question Posted:
Students also viewed these Engineering questions
-
Compounds A and B are isomeric diketones of molecular formula C6H10O2. The 1H NMR spectrum of compound A contains two signals, both singlets, at 2.2 (6 protons) and 2.8 ppm (4 protons). The 1H NMR...
-
Compounds A and B are isomeric amines of molecular formula C8H11N. Identify each isomer on the basis of the 1H NMR spectra given in Figure 22.9. Question continue over next page Figure 22.9 Compound...
-
Compounds A and B are isomeric diketones of molecular formula C6H10O2. The 1H NMR spectrum of compound A contains two signals, both singlets, at 2.2 (6 protons) and 2.8 ppm (4 protons). The 1H NMR...
-
On December 1, 2011, Lavender Manufacturing Company (a corporation) purchased another company's assets, including a patent. The patent was used in Lavender's manufacturing operations; $49,500 was...
-
Aluminumlithium alloys have been developed by the aircraft industry to reduce the weight and improve the performance of its aircraft. A commercial aircraft skin material having a density of 2.55 g/cm...
-
Assume that your father is now 50 years old, that he plans to retire in 10 years, and that he expect to live for 25 years after he retires- that is, until age 85. He wants his first retirement...
-
Identify three objects for an ATM system.
-
Precise Metals, Inc., a fast-growing company that makes metals for equipment manufacturers, has an $800,000 line of credit at its bank. One section in the credit agreement says that the ratio of cash...
-
The Sherston Brick Company manufactures a standard stone block for the building industry. The production capacity for the year is 1 0 0 , 0 0 0 standard blocks. The selling price per block is $ 1 . 6...
-
Use Figure 7-13 to estimate the compressibility (Z) of supercritical toluene at T = 651 K and P = 57.5 bar. PV RT Z= N 1.1 1.0 0.9 0.8 0.7 0.6 0.5 0.4 0.3 0.2 0.1 Bo box 80 0 0.5 Noocki + O 0 +0 +...
-
110 lb m of liquefied natural gas (LNG) is stored in a rigid, sealed 5 ft 3 vessel. Due to a failure in the cooling/insulation system, the temperature increases to 100F, which is above the critical...
-
How are fewness of firms and mutual interdependence related?
-
- Advanced accounting course tuition fees 600 - Music history course tuition fees (U of A) - Payments of premiums on life insurance required by his Bank (to cover his personal mortgage) 2,400 $1,200...
-
4. Consider the IVP t y +1 y(0) = yo- (a) Without solving, determine all values of yo for which the IVP is certain to have a unique solution. (b) Solve the IVP for all such values of yo and detemine...
-
The following graphs are derived using CAPM method based on price information of AT&T and Verizon from Jan 31, 2019 to Jan 30, 2020 from Yahoo! Finance. The solid lines are derived by running...
-
Calculate al) 8(x) (x - 3) dx a2) f (x n) sin (3) cos (3x) dx 34 a3) 26(x - 1)(x4 + 2) dx b) Let be the step function 0, x < 0 0(x) = 0.5, x = 0. 1, x > 0 Show that: 8(x) == 0 (x) dx
-
Required: (a) Prepare, on a relevant cost basis, the lowest cost estimate that could be used as the basis for a quotation. Explain briefly your reasons for using each of the values in your estimate....
-
Both the U.S. Army and the University of Wisconsin are large complex entities with numerous employees. Comment on the differences to which these organizations are decentralized, as well as variations...
-
Interview managers at three companies in your area about their use of ERP. How have their experiences been similar? What accounts for the similarities and differences?
-
In the piled raft described in Problems 14.2 and 14.3, if the design load is 345 MN, what would be the settlement? Problem 14.2 & 14.3 In a piled raft, 450 mm diameter and 13 m long piles are placed...
-
In the piled raft described in Problems 14.2 and 14.3, the ultimate load carrying capacity of the piles is estimated as 500 MN. What would be the total load on the piled raft when the piles are at...
-
Prepare a case study on the new Kingdom Tower in Saudi Arabia, which will be the tallest building in the world when completed.
-
Ivanhoe Company reports the following account balances at December 3 1 , 2 0 2 5 : Accounts payable $ 7 , 7 0 0 Accounts receivable 7 , 7 0 0 Cash 9 , 1 0 0 Intangible assets 2 1 , 9 0 0 Inventory 3...
-
Citywide Company issues bonds with a par value of $ 7 3 , 0 0 0 . The bonds mature in five years and pay 8 % annual interest in semiannual payments. The annual market rate for the bonds is 6 % ....
-
Larkspur Leasing Company agrees to lease equipment to Cullumber Corporation on January 1 , 2 0 2 5 . The following information relates to the lease agreement. 1 . The term of the lease is 7 years...

Study smarter with the SolutionInn App


