Using data in Appendix C-1, determine the van der Waals parameters a and b for each of
Question:
Using data in Appendix C-1, determine the van der Waals parameters a and b for each of the following compounds.
Appendix C-1
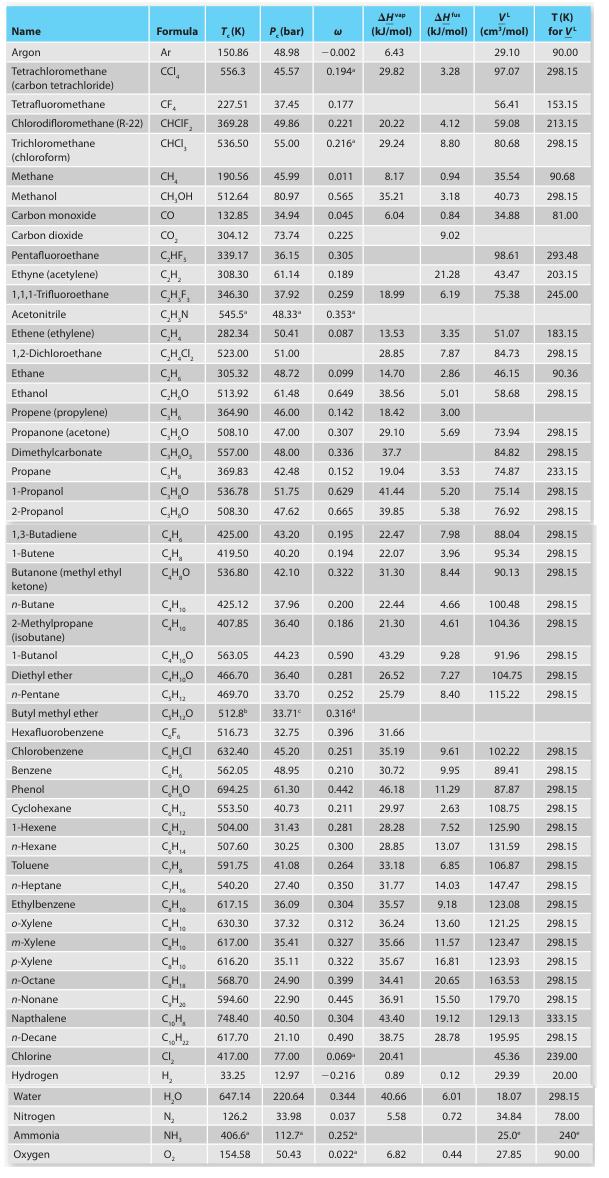
A. Methanol
B. Carbon dioxide
C. Ethanol
D. Butane
E. Octane
Transcribed Image Text:
Name Argon Tetrachloromethane (carbon tetrachloride) Acetonitrile Ethene (ethylene) 1,2-Dichloroethane. Ethane Ethanol Propene (propylene) Propanone (acetone) Dimethylcarbonate Tetrafluoromethane CF 227.51 Chlorodifloromethane (R-22) CHCIF, 369.28 Trichloromethane CHCI, 536.50 (chloroform) Methane Methanol Carbon monoxide Carbon dioxide Pentafluoroethane Ethyne (acetylene) 1,1,1-Trifluoroethane Propane 1-Propanol 2-Propanol 1,3-Butadiene 1-Butene Butanone (methyl ethyl ketone) n-Butane: 2-Methylpropane (isobutane) 1-Butanol Diethyl ether n-Pentane. Butyl methyl ether Hexafluorobenzene Chlorobenzene Benzene. Phenol Cyclohexane 1-Hexene n-Hexane Toluene n-Heptane Ethylbenzene o-Xylene. m-Xylene p-Xylene n-Octane n-Nonanel Napthalene n-Decane Formula Chlorine Hydrogen Water Nitrogen Ammonia Oxygen Ar CCI CH CH,OH CO CO₂ C₂HF, CH₂ C.H.F. C.H.N CH₂ CH.CI, CH C₂H₂O CH CHO C,H,O, CH CHO C₂H₂O CH CH CHO CH₁ CH CHO CH,O C₂H₁₂ C₂H,₂0 CF CH₂C CH CH, CHI CH T. (K) 150.86 556.3 CH₁0 CH CH₂ CH₂ C₁₂H₂z CI₂ H₂ H₂O N₂ NH, 0₂ P. (bar) 48.98 45.57 37.45 49.86 55.00 563.05 44.23 466.70 36.40 469.70 33.70 512.8⁰ 33.71° 516.73 632.40 45.20 562.05 48.95 CHO 694.25 61.30 CH₁₂ 553.50 40.73 CH,₂ 504.00 31.43 CH₁ 507.60 30.25 C₂H₂ CH 425.00 43.20 419.50 40.20 536.80 42.10 425.12 37.96 407.85 36.40 W 190.56 45.99 0.011 8.17 512.64 80.97 0.565 35.21 132.85 34.94 0.045 6.04 304.12 73.74 0.225 339.17 36.15 0.305 308.30 61.14 0.189 346.30 37.92 0.259 545.5" 48.33" 282.34 50.41 523.00 51.00 305.32 48.72 513.92 61.48 364.90 46.00 508.10 47.00 557.00 48.00 369.83 42.48 536.78 51.75 508.30 47.62 591.75 41.08 540.20 27.40 617.15 36.09 630.30 37.32 617.00 35.41 616.20 35.11 568.70 -0.002 0.194 594.60 748.40 617.70 417.00 33.25 647.14 220.64 126.2 33.98 406.6* 112.7 154.58 50.43 0.177 0.221 0.216 0.353" 0.087 0.099 0.649 0.142 0.307 0.336 0.152 0.629 0.665 0.195 0.194 0.322 0.200 0.186 0.590 0.281 0.252 0.3164 AHvap AH fus VL (kJ/mol) (kJ/mol) (cm³/mol) 6.43 29.10 29.82 97.07 20.22 29.24 0.344 0.037 0.252 0.022" 18.99 13.53 28.85 14.70 38.56 18.42 29.10 37.7 19.04 41.44 39.85 22.47 22.07 31.30 32.75 0.396 31.66 0.251 35.19 0.210 30.72 0.442 46.18 0.211 29.97 0.281 28.28 0.300 28.85 0.264 33.18 0.350 31.77 0.304 35.57 0.312 36.24 0.327 35.66 0.322 35.67 24.90 0.399 34.41 22.90 0.445 36.91 40.50 0.304 43.40 21.10 0.490 38.75 77.00 0.069 20.41 12.97 -0.216 0.89 40.66 5.58 22.44 21.30 43.29 26.52 25.79 6.82 3.28 4.12 8.80 0.94 3.18 0.84 9.02 21.28 6.19 3.35 7.87 2.86 5.01 3.00 5.69 3.53 5.20 5.38 7.98 3.96 8.44 4.66 4.61 9.28 7.27 8.40 9.61 9.95 11.29 2.63 7.52 13.07 6.85 14.03 9.18 13.60 11.57 16.81 20.65 15.50 19.12 28.78 0.12 6.01 0.72 0.44 56.41 59.08 80.68 35.54 40.73 34.88 98.61 43.47 75.38 51.07 84.73 46.15 58.68 73.94 84.82 74.87 75.14 76.92 88.04 95.34 90.13 100.48 104.36 T(K) for VL 90.00 298.15 153.15 213.15 298.15 90.68 298.15 81.00 293.48 203.15 245.00 183.15 298.15 90.36 298.15 298.15 298.15 233.15 298.15 298.15 298.15 298.15 298.15 298.15 298.15 91.96 298.15 104.75 298.15 115.22 298.15 102.22 298.15 89.41 298.15 87.87 298.15 108.75 298.15 125.90 298.15 131.59 298.15 106.87 298.15 147.47 298.15 123.08 298.15 121.25 298.15 123.47 298.15 123.93 298.15 163.53 298.15 179.70 298.15 129.13 333.15 195.95 298.15 45.36 239.00 29.39 20.00 18.07 34.84 25.0⁰ 27.85 298.15 78.00 240* 90.00
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Chemical Engineering Thermodynamics
ISBN: 9781111580704
1st Edition
Authors: Kevin D. Dahm, Donald P. Visco
Question Posted:
Students also viewed these Engineering questions
-
Van der Waals Equation and Critical Points (a) In p V- diagrams the slope p/V along an isotherm is never positive. Explain why. (b) Regions where p/V = 0 represent equilibrium between two phases;...
-
Calculate the van der Waals parameters of carbon dioxide from the values of the critical constants and compare your results with the values for and b in Table 7.4. Table 7.4 RedlichKwong van der...
-
The van der Waals equation of state is where a and b are temperature-independent parameters that have different values for each gas. For carbon dioxide, a = 0.3640 Pa m 6 mol 2 and b = 4.267 Ã...
-
solve the following instance of the 0/1 kropsack problem using dynamic programming approach. Assume that the knapsack capacity is 9. Item 1 3 4 weight 6 2 5 7 Benef $8 $4 $6 $10
-
Explain the difference between a standard costing system and the Kaizen costing system popularized in Japan.
-
Why would a mortgagor who has defaulted request the court to hold a sale of his property rather than allow the mortgagee to foreclose?
-
A snow-cone machine at an icecream shop costs \(\$ 15,000\). The machine is expected to generate profits of \(\$ 2,500\) each year of its 10-year useful life. At the end of the 10 years, the machine...
-
Derivative Transaction On January 2, 2010, Jones Company purchases a call option for $300 on Merchant common stock. The call option gives Jones the option to buy 1,000 shares of Merchant at a strike...
-
Assume the company is transitioning from a traditional file environment to a database management system / relational database. - Describe and explain the challenges / issues in transitioning from a...
-
Using data in Appendix C-1, determine the Soave parameters a and b for each of the following compounds at the temperature T = 300 K. Appendix C-1 A. Argon B. Carbon monoxide C. 1-Propanol D. Pentane...
-
Shows a general comparison between arguments a and b, where data_t, the data type of the arguments, is defined (via typedef) to be one of the integer data types listed in Figure 3.1 and either signed...
-
Janus Venture Fund, an equity fund with strong historical performance, had a return of 45 percent for the year 2000. However, it paid out $16.38 per share as a result of selling securities which had...
-
Describe the classical method to determine probability with example.
-
Outline the key features of each of the seven steps in the quantitative analysis approach.
-
In a scatter diagram, the independent variable is plotted on the a. vertical axis. b. trend line. c. horizontal axis. d. origin of the axis.
-
What are the strengths of using decision trees?
-
Explain the limitation in the use of graphical solution for an LP problem. What are its advantages, even when its specific application is not suitable?
-
In Problem 4, Dunder-Mifflin, Inc. hires an investment banker for the sale of the 600,000 bonds. The investment banker charges a fee of 2% on each bond sold. What is the cost of debt to DMI if the...
-
Suppose the index goes to 18 percent in year 5. What is the effective cost of the unrestricted ARM?
-
Use the averaging principle developed in Problem 9 to find the temperature distribution of the plate shown in Figure P10, using the 3 3 grid and the given values T a = 150C and T b = 20C. Figure P10...
-
Solve the following equations: 7x + 9y 9z = 22 + 2y 4z = 12 + 5 z %3D -2 Z.
-
The following table shows how many hours in process reactors A and B are required to produce 1 ton each of chemical products 1, 2, and 3. The two reactors are available for 35 and 40 hrs per week,...
-
What are some movements which help a customer feel welcome and heard?
-
Explain (in writing only) how the fishbone diagram can be used by a team to resolve a situation where work delays are frequently occurring. They could be due to workers, material, methods, or the...
-
Keep answers short: 2-5. Minimum Contacts. Seal Polymer Industries sold. two freight containers of latex gloves to Med-Express, Inc., a company based in North Carolina. When Med-Express failed to...

Study smarter with the SolutionInn App


