We know that many binary mixtures contain an azeotrope, which means that the liquid and vapor phases
Question:
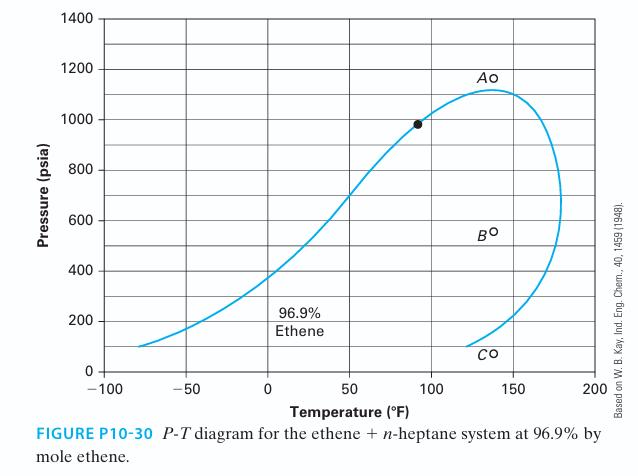
We know that many binary mixtures contain an azeotrope, which means that the liquid and vapor phases for the mixture have the same composition. Can a system have two azeotropes for a given pressure or temperature? If it exists, find a system and plot the data.
Transcribed Image Text:
Pressure (psia) 1400 1200 1000 800 600 400 200 0+ -100 -50 0 96.9% Ethene 100 Ao BO 50 Temperature (°F) FIGURE P10-30 P-T diagram for the ethene + n-heptane system at 96.9% by mole ethene. со 150 200 Based on W. B. Kay, Ind. Eng. Chem., 40, 1459 (1948).
Step by Step Answer:

This question has not been answered yet.
You can Ask your question!
Related Book For 

Fundamentals Of Chemical Engineering Thermodynamics
ISBN: 9781111580704
1st Edition
Authors: Kevin D. Dahm, Donald P. Visco
Question Posted:
Students also viewed these Engineering questions
-
The overall goal of this problem is to compute the PV and PT equilibrium diagramsfor a single component fluid described by the van derWaals equation of state. Let us recall the key things we need to...
-
Vapor?liquid equilibrium data for mixtures of acetone (A) and ethanol at 1 atm are given in the following table: (a) Use the given data to construct a Txy diagram for this system. (b) A thermocouple...
-
A binary mixture of mole fraction zj is flashed (o conditions T and P, Fur one of the following determine: the equilibrium mole fractions x1 and y1 of the liquid and vapor phases formed, the molar...
-
Q8 Question: 9 A small particle of mass m moving inside a heavy, hollow and straight tube along the tube axis undergoes elastic collision at two ends. The tube has no friction and it is closed at one...
-
Jody Hunter and Jim Boling, two managers at Georgia- Pacific Corporation, a paper manufacturer, disagree about continuing the companys membership in Business for Affordable Medicine (BAM), a...
-
(a) Repeat Prob. 11.19, but this time let the external force be a Dirac delta function: F(t) = kS(t) (for some constant k). 17 [Note that the acceleration is now discontinuous at t = 0 (though the...
-
A statistics student has constructed a confidence interval for the mean height of daughters whose mothers are 66 inches tall, and a prediction interval for the height of a particular daughter whose...
-
Allison Cooper opened Allisons Toy House. As her newly hired accountant, your tasks are to do the following: 1. Journalize the transactions for the month of October. 2. Record to subsidiary ledgers...
-
1. Provide an update on your civic activity. What are you doing? How is it going? What are you observing? Has the activity aligned with your expectations? 2. what are three potential negative...
-
Figure P10-30 is a P-T diagram for the ethene + n-heptane system at 96.9% by mole ethene (Kay, 1948). Please answer the following questions based on this figure. Note that the in the figure...
-
A separation stream off the main reactor effluent contains almost exclusively ethyl benzene, benzene, and toluene at 1 bar and 100C. You determine that the stream flow rate is made up of 34 kg/s of...
-
Consider a unit mass of \(\mathrm{N}_{2}\) in equilibrium at \(p=1 \mathrm{~atm}\) and \(T=300 \mathrm{~K}\). For these conditions, the vibrational relaxation time is \(190 \mathrm{~s}\). Assume...
-
How does Title IX enforcement procedures work? (3-prong test) What were the specifics of the Grove City decision and how does it apply to federal financial assistance to an institution? How does...
-
Windsor Company's income statement for the year ended December 31, 2025, contained the following condensed information. Service revenue $843,000 Operating expenses (excluding depreciation) $622,000...
-
Many diets recommend reducing your intake of carbohydrates. Why are carbohydrates important for cellular function from a biochemical perspective?
-
When I use the command mysql -u root -p in Codio, it asks for the password, but when I try to type a password, nothing happens, and it won't let me type anything.? Discuss
-
Top managers of Preston Industries predicted 2024 sales of 14,400 units of its product at a unit price of $10.00. Actual sales for the year were 14,000 units at $11.00 each. Variable costs were...
-
Find the predicted value of y, using each model for x = 10. a) y-hat = 2 + 0.8 ln x b) log y-hat = 5 0.23x c) 1/y-hat = 17.1 1.66x
-
Which of the following is NOT a magnetic dipole when viewed from far away? a) A permanent bar magnet. b) Several circular loops of wire closely stacked together with the same current running in each...
-
The mechanical work W done in using a force F to push a block through a distance D is W ! FD. The following table gives data on the amount of force used to push a block through the given distance...
-
Plane A is heading southwest at 300 mi/hr, while plane B is heading west at 150 mi/hr. What are the velocity and the speed of plane A relative to plane B?
-
The following table shows the hourly wages, hours worked, and output (number of widgets produced) in one week for ve widget makers. Use MATLAB to answer these questions: a. How much did each worker...
-
Two nodes, A and B, are each attempting to transmit one small frame to each other over a shared ethernet medium. Assume the signal propagation delay is zero, and a small frame takes 50 s to transmit,...
-
A seller has one item that he would like to sell. He can at any time sell it to a dealer who will pay $50 for the item. He is also considering two other ways to try to sell the item: posting a price...
-
Prepare partial income statements for 2022 through gross profit, under each of the following cost flow assumptions. (Round answers to 2 decimal places, e.g. 125.25.) (1) Specific identification...

Study smarter with the SolutionInn App


