Calculate the total coulombic potential energy of a Na + in a NaCl crystal by considering only
Question:
Calculate the total coulombic potential energy of a Na+ in a NaCl crystal by considering only up to the fourth nearest neighbors of Na+. The coulombic potential energy for two ions of opposite charges separated by a distance r is given by:
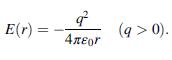
Transcribed Image Text:
E(r) = (q > 0). 4reor
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
The coulombic potential energy of a Na ion in a NaCl crystal due to its interaction with other ions ...View the full answer

Answered By

BillClinton Muguai
I have been a tutor for the past 5 years. I have experience working with students in a variety of subject areas, including computer science, math, science, English, and history. I have also worked with students of all ages, from elementary school to college. In addition to my tutoring experience, I have a degree in education from a top university. This has given me a strong foundation in child development and learning theories, which I use to inform my tutoring practices.
I am patient and adaptable, and I work to create a positive and supportive learning environment for my students. I believe that all students have the ability to succeed, and it is my job to help them find and develop their strengths. I am confident in my ability to tutor students and help them achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
By how much does the gravitational potential energy of a 64-kg pole vaulted change if his center of mass rises about 4.0m during the jump?
-
By how much does the gravitational potential energy of a 54-kg pole vaulter change if her center of mass rises about 4.0 m during the jump?
-
The electric potential energy of a uniform sphere of charge q and radius r is given by (a) Does the energy represent a tendency for the sphere to bind together or blow apart? The nuclide 239Pu is...
-
Kindly solve both the question. Thanks in advance.Question 1Question 2 Below are the transactions for Baste SewIng Shop for March, the first month of operations. March 1 Issue common stock in...
-
You are considering whether to invest in two stocks, Stock A and Stock B. Stock A has a beta of 1.15 and the standard deviation of its returns has been estimated to be 0.28. For Stock B, the beta is...
-
In the cold vulcanization of rubber, disulfur dichloride (S 2 Cl 2 ) is used as a source of sulfur atoms, and those sulfur atoms form bridges, or cross-links, between polymer chains. S 2 Cl 2 can be...
-
What happens to the membrane potential of a neuron during an action potential?
-
The accounting system for Amaldi Co. reflected the following quality costs for 2009 and 2010: a. Which of these are costs of compliance, and which are costs of noncompliance? b. Calculate the...
-
A 2 5 0 g car and a 5 0 g mass are connected by a light stringwove the foot an hte car s on a Ta ate The art tive aninitial velocity of 0 . 6 0 m / s away from the pulley. Friction between the car...
-
Ten years ago Diana Torres wrote what has become the leading Tort textbook. She has been receiving royalties based on revenues reported by the publisher. These revenues started at $1 million in the...
-
What is the full electronic configuration of Li? Since the ionization energy of Li is 5.39 eV, how much is the effective nuclear charge? What can you say about the screening of the other electrons?
-
The interaction energy between Na + and Cl - ions in the NaCl crystal can be written as where the energy is given in joules per ion pair and the interionic separation r is in meters. The numerator...
-
What major types of transactions occur in the purchasing process? What financial statement accounts are affected by each type of transaction?
-
ACME Drilling is evaluating an offshore oil-drilling platform for possible impairment. The company estimates the following: book value, $18.3 million; fair value, $12.6 million; sum of estimated...
-
Carly, your client, is Chief Accounting Office of a national retail company, which sells high end-clothing. She is considering switching inventory costing method from FIFO to LIFO. Provide the...
-
Heidi Inc, acquired a 15 year asset for $100,000. The mid-quarter convention applied to this asset, which was acquired in the second quarter. The straight line method was not elected. The company...
-
Write an equation for the acid-base reaction between 2,4-pentanedione and sodium ethoxide and calculate its equilibrium constant(Keq). The pK of 2,4-pentanedione is 9; that of ethanol is 15.9....
-
The inventory method assumes its stock has been rotated during the month is called? Explain in your words.
-
Prove that a 2 2 matrix A is reducible if and only if a12a21 = 0.
-
Maria Castigliani is head of the purchasing department of Ambrosiana Merceti, a medium-sized construction company. One morning she walked into the office and said, The main problem in this office is...
-
Write each answer with a reasonable number of figures. Find the absolute and percent relative uncertainty for each answer. (a) [12.41 (0.09) 4.16 ( 0.01)] 7.068 2 ( 0.000 4) = ? (b) [3.26 (0.10) ...
-
How many milliliters of 53.4 (0.4) wt% NaOH with a density of 1.52 ( 0.01) g/mL will you need to prepare 2.000 L of 0.169 M NaOH? (b) If the uncertainty in delivering NaOH is [1] 0.01 mL, calculate...
-
(a) How many milliliters of 53.4 (0.4) wt% NaOH with a density of 1.52 ( 0.01) g/mL will you need to prepare 2.000 L of 0.169 M NaOH? (b) If the uncertainty in delivering NaOH is [1] 0.01 mL,...
-
A company owns some beachfront property and it is now going to develop this property into a resort. However, the future demand for a resort in this location is unknown and the company is concerned...
-
How do ethical leaders foster a climate of continuous learning and ethical reflection, encouraging their teams to engage in ethical discourse, confront ethical dilemmas, and cultivate a heightened...
-
Q1. Assuming that the clay soil has a yielding function as the following: 9 (p)-p'p+ = 0 M where p' is the mean effective stress, and q is the deviatoric stress. p' is the effective preconsolidation...

Study smarter with the SolutionInn App


