A refrigerator with R-134a as the working fluid has a minimum temperature of 10C and a maximum
Question:
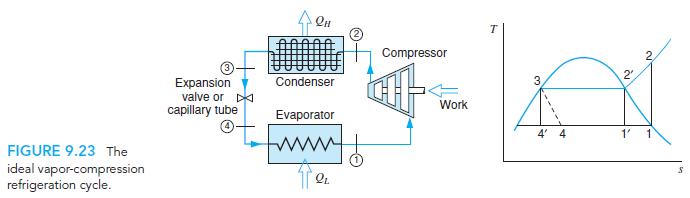
A refrigerator with R-134a as the working fluid has a minimum temperature of −10◦C and a maximum pressure of 1 MPa. Assume an ideal refrigeration cycle, as in Fig. 9.23. Find the specific heat transfer from the cold space and that to the hot space, and the COP.
Transcribed Image Text:
Qu T Compressor 2. Expansion valve or D capillary tube Condenser Work Evaporator 4' 4 FIGURE 9.23 The ideal vapor-compression refrigeration cycle.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
Exit evaporator sat vapor 10C from B51 h 1 39228 s 1 173...View the full answer

Answered By

Bombothula Navya Laxmi
I'm responsible for helping students learn and understand new concepts
I'm able to growth mindset toward student learning and teaching practice
I have a good knowledge in my subjects so that can able give you answers.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Thermodynamics
ISBN: 9781118131992
8th Edition
Authors: Claus Borgnakke, Richard E. Sonntag
Question Posted:
Students also viewed these Sciences questions
-
A refrigerator with R-12 as the working fluid has a minimum temperature of 10C and a maximum pressure of 1 MPa. Assume an ideal refrigeration cycle as in Fig. 11.32. Find the specific...
-
A refrigerator with R-12 as the working fluid has a minimum temperature of 10C and a maximum pressure of 1 MPa. The actual adiabatic compressor exit temperature is 60C. Assume no pressure loss in the...
-
A gas turbine with air as the working fluid has two ideal turbine sections, as shown in Fig. P11.53, the first of which drives the ideal compressor, with the second producing the power output. The...
-
The Sooner Equipment Company has total assets of $100 million. Of this total, $40 million was financed with common equity and $60 million with debt (both long- and short-term). Its average accounts...
-
Why must Eurobanks operate with narrower spreads than domestic banks? What would happen if the spreads were equal in both markets?
-
Describe the intersection of the horizontal plane z = h and the hyperboloid x 2 4y 2 + 4z 2 = 1. For which values of h is the intersection empty?
-
Why are cohesion and adhesion important to water transport in plants?
-
Starbucks Coffee Co. uses a data based approach to improving the quality and customer satisfaction of its products. When survey data indicated that Starbucks needed to improve its package sealing...
-
Your organization has noticed an alarming increase in injury incident reports this winter. The CEO has asked you to write a report summarizing the common incidents reported so far. What kind of...
-
The Dud Company purchases raw materials on terms of "2/10, net 30." A review of the company's records by the owner, Ms. Dud, revealed that payments are usually made 15 days after purchases are...
-
A refrigeration cycle, as in Fig. 9.23, can be used for cooling or for heating purposes using one of the two heat exchangers. Suppose a refrigerator should cool meat at 10C in a 30C kitchen. What are...
-
Can the combined cycles in the previous problem deliver more heat than what comes from the R-410a? Find any amounts, if so, by assuming some conditions. Data from previous problem A small utility...
-
Solve each equation, and check the solution. If applicable, tell whether the equation is an identity or a contradiction. -2x - 3(4 - 2x) = 2(x - 3) + 2
-
Ensure your response is positive and appropriate and that the children will learn about how their actions affect others without feeling ashamed or humiliated Susan(4yrs 2mnths) and Rachel (4yrs...
-
The children had created a tower with the blocks the previous day and called them "the Airport control tower". The play was kept so children could continue their play. Today, Alex (4.5yrs) was alone...
-
If 3(n-8)-n+1 = 1, what is the value of n?
-
Using Cash flow sweep assumption to pay down revolver and senior debt, what is the senior bank debt at the end of the year 2021 assuming that: Revolver balance at the beginning of the year = $200...
-
Porter Corporation makes and sells a single product called a Yute. The company is in the process of preparing its Selling and Administrative Expense Budget for the last quarter of the year. The...
-
A beam of light in air is incident on a stack of four flat transparent materials with indices of refraction 1.20, 1.40, 1.32, and 1.28. If the angle of incidence for the beam on the first of the four...
-
Akramin just graduated with a Master of Engineering in Manufacturing Engineering and landed a new job in Melaka with a starting salary of RM 4,000 per month. There are a number of things that he...
-
Propose an efficient synthesis for each of the following compounds using the malonic ester synthesis. (a) (b) (c) (d) (e)
-
Starting with diethyl malonate and using any other reagents of your choice, proposean efficient synthesis for each of the following compounds: (a) (b) (c) .
-
The malonic ester synthesis cannot be used to make 2,2-dimethylhexanoic acid. Explain why not.
-
Dana is a surviving spouse. Her husband Jim passed away on June 14th, 2021. Dana works as a teacher at a local high school. She paid $435 in costs for school supplies out of her own pocket. She also...
-
calculate the consideration for 75% stock and 5% cash ,50% stock and 50% cash, 0% stock and 100% cash Figure 6. SunTrust financials as of March 31, 2004. Balance Sheet Total Assets ($000)...
-
Palacios Incorporated estimates that its retained earnings break point (BPRE) is $45 million, and its WACC is 14.8 percent if common equity comes from retained earnings. However, if the company...

Study smarter with the SolutionInn App


