A rigid tank with 0.5 kg ammonia at 1600 kPa, 160C is cooled in a reversible process
Question:
A rigid tank with 0.5 kg ammonia at 1600 kPa, 160◦C is cooled in a reversible process by giving heat to a reversible heat engine that has its cold side at ambient temperature, 20◦C, shown in Fig. P6.186. The ammonia eventually reaches 20◦C and the process stops. Find the heat transfer from the ammonia to the heat engine and the work output of the heat engine.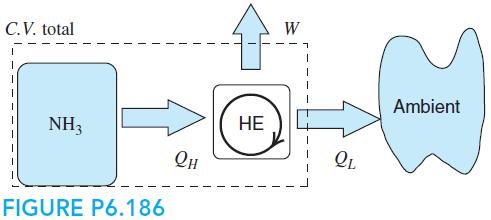
Transcribed Image Text:
C.V. total W Ambient NH3 НЕ Он FIGURE P6.186
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
We can use the First Law of Thermodynamics to find the heat transfer and work outp...View the full answer

Answered By

BillClinton Muguai
I have been a tutor for the past 5 years. I have experience working with students in a variety of subject areas, including computer science, math, science, English, and history. I have also worked with students of all ages, from elementary school to college. In addition to my tutoring experience, I have a degree in education from a top university. This has given me a strong foundation in child development and learning theories, which I use to inform my tutoring practices.
I am patient and adaptable, and I work to create a positive and supportive learning environment for my students. I believe that all students have the ability to succeed, and it is my job to help them find and develop their strengths. I am confident in my ability to tutor students and help them achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Thermodynamics
ISBN: 9781118131992
8th Edition
Authors: Claus Borgnakke, Richard E. Sonntag
Question Posted:
Students also viewed these Sciences questions
-
A rigid tank with 3 kg of H2O at 150 kPa, x = 0.2 is heated with 1000 kJ. Determine (a) The final pressure. (b) Phase composition of H2O.
-
A rigid tank with a volume of 2.5 m3 contains 15 kg of saturated liquidvapor mixture of water at 75C. Now the water is slowly heated. Determine the temperature at which the liquid in the tank is...
-
A rigid tank with a volume of 1.8 m3 contains 15 kg of saturated liquid-vapor mixture of water at 90oC. Now the water is slowly heated. Determine the temperature at which the liquid in the tank is...
-
Draw the BST that results when you insert the keys E A S Y QUE S T I O N in that order into an initially empty tree. What is the height of the resulting BST?
-
The shaft of a stepper motor is directly connected to a lead screw that drives a worktable in an x-y positioning system. The motor has a step angle = 5(. The pitch of the lead screw is 6 mm, which...
-
Antonio Ltd has planned production and sales for the next nine months as follows: During the period, the business plans to advertise so as to generate these increases in sales. Payments for...
-
The median is different from 120. An analysis of the data reveals that there are 35 minus signs and 28 plus signs. Use the sign test to test the given alternative hypothesis at the \(\alpha=0.05\)...
-
Trendy T-Shirt Factory manufactures plain white and solid-colored T-shirts. Inputs include the following: Additionally, the colored T-shirts require 3 ounces of dye per shirt at a cost of $ 0.40 per...
-
Observe changes occurred in value of all registers, which is accessed by operand in Debug Mode, then fill the blanks. (Write all esi value in L1 and ebx value in L2) TITLE Practice08-1 INCLUDE...
-
Sound Investments, Inc. is a large retailer of stores equipment. The controller is about to prepare the budget for the first quarter of 20x2. Past experience has indicated that 75 percent of the...
-
Asmall halogen light bulb receives electrical power of 50W. The small filament is at 1000 K and gives out 20% of the power as light and the rest as heat transfer to the gas, which is at 500 K; the...
-
Two tanks contain steam and they are both connected to a piston/cylinder, as shown in Fig. P6.182. Initially the piston is at the bottom, and the mass of the piston is such that a pressure of 1.4 MPa...
-
Write a plan for proof for one case of the Composition Theorem. Q Q'e R' R P R" m Q"
-
Could you provide an example of a societal reaction or response to instances of crime and deviant behavior?
-
Your firm needs to pay its French supplier 458,000. If the exchange rate is $0.68/$?, how many dollars will you need to make the exchange?
-
Gray Uniforms is a wholesaler who sells school uniforms to retailers. On August 1, Gray contracts with Excel School Uniforms to sell 2,000 uniforms to Excel to be delivered September 1. The contract...
-
A young girl from the Kayan wears a neck ring made of brass. it looks as if there are 21 individual rings but the ring is actually one continuous length of brass fashioned (bent) into a coil. the...
-
The Down and Out Co. just issued a dividend of $1.10 per share on its common stock. The company is expected to maintain a constant 0.03 growth rate in its dividends indefinit ely. If the stock sells...
-
The Securities and Exchange Commission (SEC) is an important governmental organization that exists primarily for the protection of the interests of investors in the U.S. securities markets. The SEC...
-
The Adjusted Trial Balance columns of a 10-column work sheet for Webber Co. follow. Complete the work sheet by extending the account balances into the appropriate financial statement columns and by...
-
Using the data in the following table, predict the sign and magnitude of ÎH° for each of the following reactions. In each case, identify whether the reaction is expected to be endothermic...
-
For each of the following processes predict the sign of ÎS for the reaction. In other words, will ÎS sys be positive (an increase in entropy) or negative (a decrease in entropy)? a. b. c....
-
At room temperature, molecules spend most of their time in lower energy conformations. In fact, there is a general tendency for any system to move toward lower energy. As another example, electrons...
-
You are interested in clinical care, as well as public health. I need to make a choice, you think to yourself. "Not necessarily," your advisor says. "There are many ways to combine a clinical care...
-
select any three project management principles and discuss their implications and impact in your organizational context. select any two project performance domains and discuss their practical...
-
Engine power is sometimes expressed in terms of "horsepower." One horsepower was defined by James Watt, who observed that a horse could walk 3.3 km/hr while exerting a nearly constant force of 816 N....

Study smarter with the SolutionInn App


