Two rigid tanks, shown in Fig. P6.97, each contains 10 kg N 2 gas at 1000 K,
Question:
Two rigid tanks, shown in Fig. P6.97, each contains 10 kg N2 gas at 1000 K, 500 kPa. They are now thermally connected to a reversible heat pump, which heats one tank and cools the other, with no heat transfer to the surroundings. When one tank is heated to 1500 K, the process stops. Find the final (P, T) in both tanks and the work input to the heat pump, assuming constant heat capacities.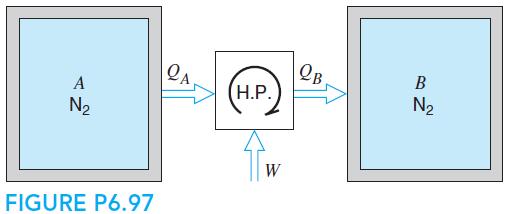
Transcribed Image Text:
OB H.P. A N2 N2 W FIGURE P6.97
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
Given the following parameter P A 1 P B1 T A1 T B1 T B2 Find the answers to these missing parameters ...View the full answer

Answered By

Saida kasyoka
I am an experienced writer with four years of work experience Over the course of four years, as a writer, I have completed more than 200 papers. I was pursuing a Diploma in Electrical/Electronics Engineering prior to becoming an academic/article writer. My favorite subjects to write about for essays are sociology, literature, psychology, political science, ethics, religion, and the English language. I'm very passionate about tutoring.
I enjoy looking for original ways to offer information, which is why I love tutoring. My audience and my aim will both influence the tone. Any tutoring I do should always have an objective—what I aim to achieve. Knowing who my audience is should be obvious from knowing what my purpose is, and those two things should be all I need to know what to offer my student.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Thermodynamics
ISBN: 9781118131992
8th Edition
Authors: Claus Borgnakke, Richard E. Sonntag
Question Posted:
Students also viewed these Sciences questions
-
Two rigid tanks each contain 10 kg N2 gas at 1000 K, 500 kPa. They are now thermally connected to a reversible heat pump, which heats one and cools the other with no heat transfer to the...
-
A mass-loaded piston/cylinder, shown in Fig P6.133, containing air is at 300 kPa, 17C with a volume of 0.25 m3, while at the stops V = 1 m3. An air line, 500 kPa, 600 K, is connected by a valve that...
-
A reversible heat pump uses 1 kW of power input to heat a 25oC room, drawing energy from the outside at 15oC. Assuming every process is reversible, what are the total rates of entropy into the heat...
-
Data-2-Go manufactures and sells flash drives. The company produces only when it receives orders and, therefore, has no inventories. The following information is available for the current month:...
-
"Monetary disequilibrium leads to balance of payments problems under fixed exchange rates, and a currency problem under floating exchange rates." Discuss this statement with reference to the monetary...
-
Discuss IIQM.
-
In a science museum, you may have seen a Foucault pendulum, which is used to demonstrate the rotation of the earth. In one museums pendulum, the 110 kg bob swings from a 15.8-m-long cable with an...
-
Budgeting and ethics. Delma Company manufactures a variety of products in a variety of departments, and evaluates departments and departmental managers by comparing actual cost and output relative to...
-
A financial institution is processing transactions and wishes to improve its security posture. The institution divides its network into different sections to minimize risk while actively updating or...
-
A helical compression spring has a scale of 400lbs/in, an inside diameter of 2.25 inches, a free length of 8 inches, and squared and ground ends. The material is to be chrome vanadium steel. For a...
-
Two rigid, insulated tanks are connected with a pipe and valve. One tank has 0.5 kg air at 200 kPa, 300K and the other has 0.75 kg air at 100 kPa, 400 K. The valve is opened, and the air comes to a...
-
A hydrogen gas in a piston/cylinder assembly at 300 K, 100 kPa with a volume of 0.1 m 3 is now slowly compressed to a volume of 0.01 m 3 while cooling in a reversible isothermal process. What is the...
-
Represent each complex number graphically and give the rectangular form of each. 18.3 540.0
-
Shown to the right are speed- time graphs for two carts that were involved in a collision similar to what you observed in Experiment 2 of this activity. (To remind you, you gave the "launched cart" a...
-
Explain the steps authitor can take to protect themselves against failure to detect fraud.
-
Harold is a commercial lines underwriting manager with Insurance Company (IC). Selina, a top-notch member of Harold's underwriting unit, is married to Martin, an insurance producer at a brokerage...
-
The federal return provides for a business credit called the Qualified Business Income Deduction (QBID). Does CA conform to this credit and if not, what do you do?
-
Samuel Jenkins made two investments; the first was 13 months ago and the second was two months ago. He just sold both investments and has a capital gain of $4,000 on each. If Samuel is single and has...
-
Sketch a ray diagram to show that when an object is placed at twice the focal length from a converging lens, the image formed is inverted, real, and the same size as the object.
-
Determine the annual percentage yield for a loan that charges a monthly interest rate of 1.5% and compounds the interest monthly.
-
By finding appropriate half-cell reactions, calculate the equilibrium constant at 298.15 K for the following reactions: a. 4NiOOH(s) + 2 2 O(l) 4Ni(OH) 2 (s) + O 2 (g) b. 4NO 3 (aq)+ 4H + (aq)...
-
The cell potential E for the cell Pt(s)|H 2 (g, a H2 = 1) H + (aq, a H+ = 1)NaCl(aq, m = 0.300) AgCl(s) Ag(s) is +0.260 V. Determine Cl assuming that = Na+ = Cl .
-
The Edison storage cell is described by Fe(s) FeO(s) KOH(aq, a KOH ) Ni 2 O 3 (s) NiO(s) Ni(s) and the half-cell reactions are as follows: Ni 2 O 3 (s) + H 2 O(l) + 2e 2NiO(s) + 2OH ...
-
compare the certificate of deposit (CD) of at least three banks. What factors affect certificate of deposit rates? What is the difference between a compound interest and a simple interest? ( chase,...
-
(a) In no more than two paragraphs, please explain the main differences between preferred stocks and common stocks? (b) As discussed in class, and in one paragraph, please write out what you need to...
-
VonTesler Enterprises Inc. provides equipment and service solutions to a variety of small and midsized business customers throughout the northeastern United States. VonTesler Enterprises Inc. is...

Study smarter with the SolutionInn App


