A portion of the standard electrode potential diagram of selenium is given below. What is the E
Question:
A portion of the standard electrode potential diagram of selenium is given below. What is the E° value for the reduction of H2SeO3 to H2Se in 1 M acid?
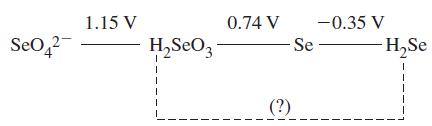
Transcribed Image Text:
Se04² 1.15 V H₂S₂O3 0.74 V -0.35 V -Se- (?) H₂Se 1 1 I T 1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
To calculate the E value for the reduction of H2SeO3 to H2Se in ...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
The reaction taking place in an electrochemical cell under standard conditions is Fe 2+ (aq) + Ag + (aq) Fe 3+ (aq) + Ag(s) a. Write two half-equations for this reaction. For each, state whether...
-
The U.S. Department of Energy's Fuel Economy Guide provides fuel efficiency data for cars and trucks. A portion of the data for 311 compact, midsized, and large cars follows. The Class column...
-
The Department of Energy and the U.S. Environmental Protection Agencys 2012 Fuel Economy Guide provides fuel efficiency data for 2012 model year cars and trucks (Department of Energy website, April...
-
Use the data in Exercise 13.28.To familiarize yourself with recursive least squares, estimate the savings functions for 19701981, 19701985, 19701990, and 19701995. Comment on the stability of...
-
Which one of the following is not an example of inventory? a. Cranes at a construction site b. Books on the shelves of a bookstore c. Apples in a supermarket d. Screws to be used in assembling tables...
-
Are the unbiased expectations and liquidity premium theories explanations for the shape of the yield curve completely independent theories? Explain why or why not.
-
This case deals with several issues regarding contract formation under the UCC. Logan and Kanawha Coal agreed to purchase coal from Detherage via a fax dated March 9, 2010. The fax stated that it had...
-
Two companies, Energen and Hastings Corporation, began operations with identical balance sheets. A year later, both required additional manufacturing capacity at a cost of $50,000. Energen obtained a...
-
Provide a clear definition of SES (Socioeconomic Status), encompassing its components such as income, education, and occupation. Discuss the societal implications of SES and its role in shaping...
-
Although relatively rare, all of the following compounds exist. Based on what you know about related compounds (for example, from the periodic table), propose a plausible name or formula for each...
-
Suppose that the sulfur present in seawater as SO 4 2 (2650 mg L 1 ) could be recovered as elemental sulfur. If this sulfur were then converted to H 2 SO 4 , how many cubic kilometers of seawater...
-
Explain why the slope of a secant line can be interpreted as an average rate of change.
-
Go to the Web. Locate the United States Code (hint: Try the Legal Information Institute). Locate Title 18, Crimes and Criminal Procedure. Locate Chapter 115, Treason, Sedition, and Subversive...
-
In 2016, the British Open awarded about $8.5 million in prize money, while the British Womens Open awarded only $3 million. Is this evidence of gender discrimination in golf? Why or why not?
-
A bag contains 23 copper rings and 7 gold rings. You reach into the bag and grab 9 rings. What is the probability that at most 1 of them is a gold ring?
-
Use the circular-flow diagram to explain how an increase in the amount of money spent by households results in an increase in the number of jobs in the economy. Describe in words what the...
-
Laura Lightfingers steals a package of filet mignon priced at $19.99 from the meat counter in a supermarket. As she leaves the store, she is approached by a security guard. Lightfingers kicks and...
-
Rick and Debbie Siravo own a beachfront home in Wrightsville Beach, N.C. During the year, they rent it for 20 weeks (140 days) at $1,100 per week and use it 10 days for personal purposes. Rick...
-
In a nonmagnetic medium, E = 50 cos (10 9 t 8x) a y + 40 sin (10 9 t 8x) a z V/m find the dielectric constant r and the corresponding H.
-
Wells Fargo & Company, headquartered in San Francisco, is one of the nations largest financial institutions. It reported the following selected accounts (in millions) as of December 31, 2009....
-
The following stockholders equity accounts, arranged alphabetically, are in the ledger of Patel Corporation at December 31, 2012. Common Stock ($2 stated value) ..................$1,600,000 Paid-in...
-
The following accounts appear in the ledger of Sather Inc. after the books are closed at December 31, 2012. Common Stock (no-par, $1 stated value, 400,000 shares authorized, 250,000 shares issued)...
-
1. What do you think Genzyme should do about its financial strategy? Answer this question from the perspective of Henri Termeer and Genzyme.
-
Mandy inherited stock upon the death of her uncle. Mandy's uncle had only owned the stock for 6 months before he died. He had a basis of $6,000 in the stock. At the date of death, the stock was...
-
1. What is defined benefit pension and how is it different from a defined contribution pension? Provide some analysis on the pros and cons. 2. Calculate the present value of the Pension liabilities...

Study smarter with the SolutionInn App


