Assume that all reactants and products are in their standard states, and use data from Table 19.1
Question:
Assume that all reactants and products are in their standard states, and use data from Table 19.1 to predict whether a spontaneous reaction will occur in the forward direction in each case.
Table 19.1
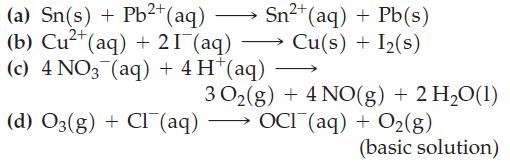
Transcribed Image Text:
(a) Sn(s) + Pb²+ (aq) (b) Cu²+ (aq) + 21 (aq) (c) 4 NO3 (aq) + 4 H*(aq) (d) O3(g) + Cl(aq) Sn²+ (aq) + Pb(s) Cu(s) + I₂(s) 30₂(g) + 4 NO(g) + 2 H₂O(1) OCI (aq) + O₂(g) (basic solution)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
To determine whether a reaction is spontaneous in the forward direction we can use standard reductio...View the full answer

Answered By

Munir Ahmed Jakhro
I am professional Tutor of of Business Courses, I did my four years Bachelor Degree from one of the Top Business schools of World "Institute of Business Administration" in year 2013. Since then I have been working as Tutor of Accounting, Finance tutor on different online platforms like this website. I am have experience of 6 years teaching business courses to students online and offline my professional job at national savings also helped me in accounting understanding .
4.90+
8+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Which of the following reactions occur spontaneously, and which can be brought about only through electrolysis, assuming that all reactants and products are in their standard states? For those...
-
Write an equation to represent the oxidation of Cl - (aq) to Cl 2 (g) by PbO 2 (s) in an acidic solution. Will this reaction occur spontaneously in the forward direction if all other reactants and...
-
According to Le Chteliers principle and the dependence of free energy on reactant and product concentrations, which statement is true? (Assume that both reactants and products are gaseous.) (a) A...
-
Two stocks, A and B, have beta coefficients of 0.8 and 1.4, respectively. If the expected return on the market is 10 percent and the risk-free rate is 5 percent, what is the risk premium associated...
-
Using the following information, compute cash flow from operating activities under (1) the U.S. approach and (2) the U.K. approach. (a) Cash paid to purchase inventory . . . . . . . . . . . . . . . ....
-
Mayville is a rural village with a population of 900. Until now, Mayville was served by a bookmobile from a larger town. The Mayville Village Council has authorized funds for a small public library,...
-
In a contract dispute between a US company and a Canadian company, the contract itself referred to provisions of the Uniform Commercial Code. Do these references alone preempt the contract from being...
-
An equimolar liquid mixture of benzene and toluene is separated into two product streams by distillation. A process flowchart and a somewhat oversimplified description of what happens in the process...
-
Problem 5. [20 pts] Verify Green's Theorem for the following vector field and region D. Namely, compute both, the line integral and the double integral. x1 1. (10pts) F (1,-1). and D is the square...
-
Ni 2+ has a more positive reduction potential than Cd 2+ . (a) Which ion is more easily reduced to the metal? (b) Which metal, Ni or Cd, is more easily oxidized?
-
For the reduction half-cell reactions Hg 2 2+ (aq) + 2 e 2 Hg(l), E = 0.797 V. Will Hg(l) react with and dissolve in HCl(aq)? in HNO 3 (aq)? Explain.
-
From the comparative balance sheet of Hata Corporation in Figure 22.12, do the following: (a) Prepare a horizontal analysis of each item for the amount of increase or decrease as well as the percent...
-
Discussion: IntroductionsDiscussion Topic Welcome to CS 230: Operating Systems! One of the best ways to build community in an online course is to get to know your fellow classmates by sharing...
-
How did the court rule on the question of whether Dr. Dawson had the authority to offer a permanent contract?
-
Question: My ATM (100 points) To answer this question, please create an application called MyATM which includes the following features: 1) The ATM should have an "Account" label and showing a...
-
Discussion questions: With reference to the reading, explain Frank Jackson's view of qualia. Do you agree with him? Why (not)? Do you believe that there are any non-physical entities, i.e.,...
-
Making a simple web application using Netbeans. 1. Create: A user (e.g. customer, staff) can sign up online (full name, email [as a username], password, phone) A registered user access logs (user...
-
In 2011, the controller of Sytec Corporation discovered that $42,000 of inventory purchases were incorrectly charged to advertising expense in 2010. In addition, the 2010 year-end inventory count...
-
Solve for the equilibria of the following discrete-time dynamical systems Pr pt+1 = Pr+2.0(I-Pr)
-
Blanco Metal Company produces the steel wire that goes into the production of paper clips. In 2010, the first year of operations, Blanco produced 50,000 miles of wire and sold 45,000 miles. In 2011,...
-
Gagliano Company has decided to introduce a new product. The new product can be manufactured by either a capital-intensive method or a labor-intensive method. The manufacturing method will not affect...
-
The condensed income statement for the Terri and Jerri partnership for 2010 is as follows. A cost behavior analysis indicates that 75% of the cost of goods sold are variable, 50% of the selling...
-
1. The wall of a refrigerated van is made of steel sheet, glass wool and plywood. The outer surface is made up of Ls = 5mm steel sheet (Ks=25 W/mK) and the inner surface is made up of Lp=15mm plywood...
-
M20 = -22.0,38.0,-68.0 iN-m, jN. m, kN.m Submit Previous Answers Consider the pipe assembly in (Figure 1). F = {30i +20j - 30k} N. F = {60i - 10j - 25k} N. Correct Figure 0.8 m F F2 Part C Add these...
-
1. There are some mistakes in the following drawing. Please point out them, and propose improvements. Then draw the improved one.

Study smarter with the SolutionInn App


