At 298 K, f G[CO(g)] = -137.2 kJ mol -1 and K = 6.5 x 10
Question:
At 298 K, ΔfG°[CO(g)] = -137.2 kJ mol-1 and K = 6.5 x 1011 for the reaction CO(g) + Cl2(g) ⇌ COCl2(g). Use these data to determine ΔfG°[COCl2(g)], and compare your result with the value in Appendix D.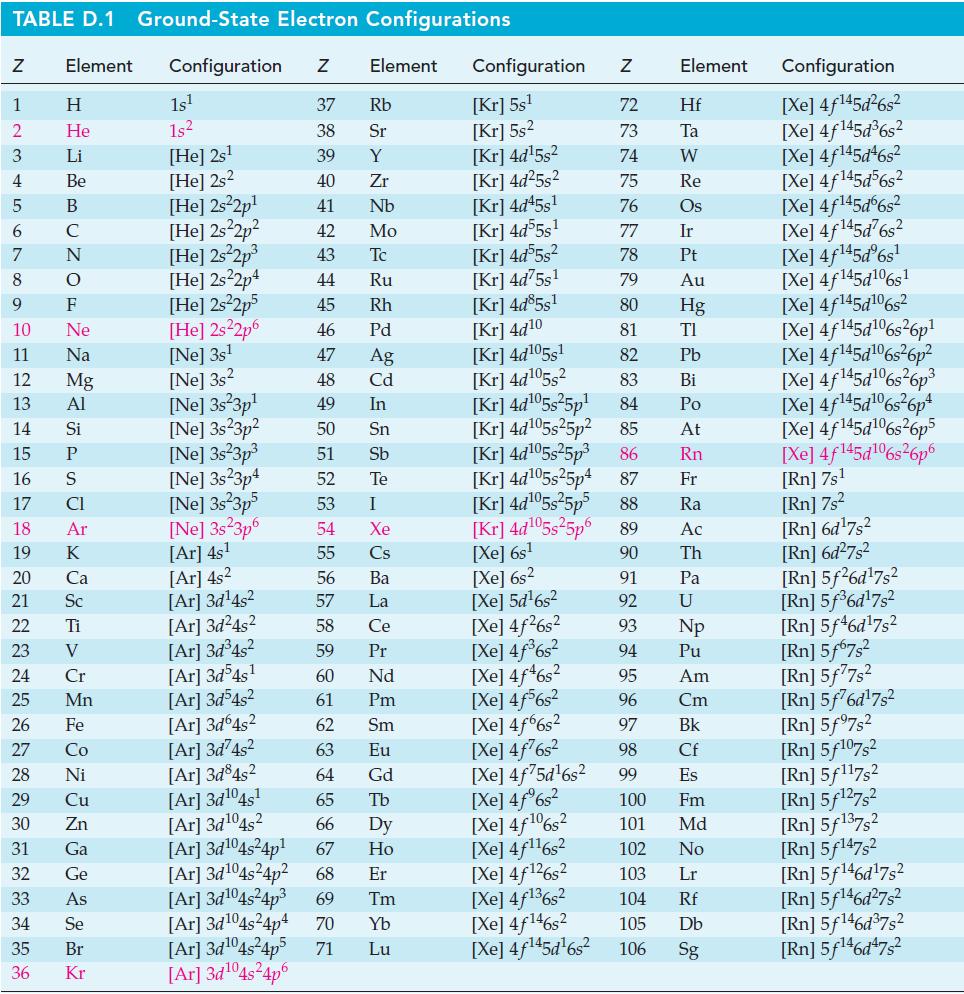
Transcribed Image Text:
TABLE D.1 Ground-State Electron Configurations Element Configuration Z Z 1 2 3 4 5 6 7 8 9 HIG&LUZON SJE ZE> 0 ≤ 2 8 2 3 5 3 3 2 2 5 2 He 10 11 12 13 14 15 16 17 18 19 20 21 Sc 22 23 24 25 Mn Mg 26 27 28 Ni 29 30 31 32 33 34 35 36 Zn Ga Ge 1s¹ 1s² [He] 2s¹ [He] 2s2 [He] 2s²2p¹ [He] 2s²2p² [He] 2s²2p³ [He] 2s22p4 [He] 2s²2p5 [He] 2s²2p6 [Ne] 3s¹ [Ne] 3s2 [Ne] 3s 3p¹ [Ne] 3s23p² 37 Rb 38 Sr 39 Y 40 Zr 41 Nb 42 Mo 43 Tc 44 Ru 45 Rh 46 Pd 47 Ag 48 Cd 49 In 50 Sn 51 Sb 52 Te 53 54 55 56 57 58 59 60 61 62 [Ar]3d²4s² 63 [Ar] 3d845² 64 [Ar]3d¹04s¹ 65 [Ar] 3d¹04s2 66 67 [Ar]3d¹04s²4p¹ [Ar] 3d¹04s²4p² 68 Er [Ar] 3d¹04s²4p³ 69 Tm 70 Yb Lu [Ne] 3s²3p³ [Ne] 3s23p4 [Ne] 3s²3p5 [Ne] 3s 3p6 [Ar] 4s¹ [Ar] 4s² [Ar] 3d¹4s² [Ar] 3d²4s² [Ar] 3d³4s² [Ar]3d54s¹ [Ar] 3d³4s² [Ar] 3d64s² Element [Ar] 3d¹04s²4p4 [Ar] 3d¹04s²4p5 71 [Ar] 3d¹04s²4p6 I Xe Cs Ba La Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Configuration Z [Kr] 5s¹ [Kr] 5s² [kr] 4d¹5s² [Kr] 4d²5s² [Kr] 4d45s¹ [kr] 4d55s¹ [kr] 4d55s² [Kr] 4d75s¹ [Kr] 4d85s1 [Kr] 4d10 [Kr] 4d105s1 [kr] 4d¹05s² [Kr] 4d¹05s²5p¹ [kr] 4d¹05s25p² [kr] 4d¹05s²5p³ [kr] 4d¹05s25p4 [Xe] 6s² [Xe] 5d¹6s² [Xe] 4f²6s² [Xe] 4f³6s² [Xe] 4f46s2 [Xe] 4f6s2 [Xe] 4f6s2 [Xe] 4f²6s² [Xe] 4f75d¹6s² [Xe] 4f%s2 [Xe] 4f106s2 [Xe] 4f¹¹6s² NRNKERKR [Xe] 4f126s2 [Xe] 4f136s2 [Xe] 4f146s2 [Xe] 4f¹45d¹6s² 72 Hf 73 Ta W 74 75 Re 76 Os 77 Element 78 79 80 81 82 83 84 85 86 87 Fr [Kr] 4d¹05s²5p5 88 Ra [Kr] 4d¹05s²5p6 89 Ac [Xe] 6s¹ 90 Th 91 92 93 94 95 96 97 98 99 Ir Pt Au Hg TI Pb Bi Po At Rn Pa U Np Pu Am Cm Bk Cf Es 100 Fm 101 Md 102 No 103 Lr 104 Rf 105 Db 106 Sg Configuration [Xe] 4f¹45d²6s² [Xe] 4f145d³6s² [Xe] 4f145d46s2 [Xe] 4f145d56s2 [Xe] 4f145d6s2 [Xe] 4f¹45d²6s² [Xe] 4f¹45dº6s¹ [Xe] 4f145d106s1 [Xe] 4f145d106s2 [Xe] 4f145d6s26p* [Xe] 4f145d106s36p? [Xe] 4f145d16s?6p3 [Xe] 4f145d6s®6p* [Xe] 4f145d16s26p5 [Xe] 4f145d106s26p6 [Rn] 7s¹ [Rn] 7s² [Rn] 6d¹7s² [Rn] 6d²7s² [Rn] 5f26d¹7s² [Rn] 5f³6d¹7s² [Rn] 5f46d¹7s2 [Rn] 5f67s² [Rn] 5f77s² [Rn] 5f76d¹7s² [Rn] 5f97s2 [Rn] 5f107,2 [Rn] 5f117s2 [Rn] 5f¹27s² [Rn] 5f137,2 [Rn] 5f147s2 Rn] 5f¹46d¹7s² [Rn] 5f¹46d²7s² [Rn] 5f¹46d³7s2 [Rn] 5f¹46d¹7s²
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
To determine fG for the reaction COg Cl2g COCl2g using the given information you can use the relatio...View the full answer

Answered By

HARSH RANJAN
Taken classes at college to graduates, Also worked as an expert to a freelancer online question-solving portal for more than 8 months with an average rating greater than 4.2 out of 5.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Use the data in Appendix 3 to calculate the equilibrium constant for the reaction Agl(s) Ag+(aq) + I2(aq) at 25C. Compare your result with the Ksp value in Table 16.2.
-
In Chapters 2 (Section 2.8) and 3 (Section 3.7) we analyzed the risk and return of the Orange County portfolio, using the building blocks of zero coupon bonds and duration in this exercise, we...
-
A tabulation of more precise thermodynamic data than are presented in Appendix D lists the following values for H 2 O(l) and H 2 O(g) at 298.15 K, at a standard state pressure of 1 bar. (a) Use these...
-
The United Arab Emirates (UAE) is unique in that expatriates constitute more than 80 per cent of the population. As the country continues to grow and accept foreigners at an astounding pace, Emiratis...
-
Fuqua Companys sales budget projects unit sales of part 198Z of 10,000 units in January, 12,000 units in February, and 13,000 units in March. Each unit of part 198Z requires 4 pounds of materials,...
-
Draw the shear and moment diagrams for the double overhangbeam. 10 kN 5 kN 5 kN 2m+2. -2 m2m 2 m
-
Which of the following is an activity not usually associated with forensic accounting and fraud examination consulting and litigation support? 1. A. Assessing fraud risk associated with internal...
-
Yost-Perry Industries (YPI) manufactures a mix of affordable guitars (A. B, C) that are fabricated and assembled at four different processing stations (W, X, Y, Z). The operation is a batch process...
-
Direct labor-hours Machine-hours Total fixed manufacturing overhead cost Variable manufacturing overhead per machine-hour Variable manufacturing overhead per direct labor-hour Required: 1. Compute...
-
At 1000 K, an equilibrium mixture in the reaction CO 2 (g) + H 2 (g) CO(g) + H 2 O(g) contains 0.276 mol H 2 0.276 mol CO 2 , 0.224 mol CO, and 0.224 mol H 2 O. (a) What is K at 1000 K? (b)...
-
Why is r G such an important property of a chemical reaction, even though the reaction is generally carried out under nonstandard conditions?
-
The wages payable and wages expense accounts at October 31, after adjusting entries have been posted at the end of the first month of operations, are shown in the following T accounts: Determine the...
-
In decision theory, probabilities are associated with a. payoffs. b. alternatives. c. states of nature. d. none of the above.
-
What is the relationship between airline fixed costs and marginal costs and how is it related to revenue management?
-
A machine is supposed to fill soft drink cans to 12 ounces. It appears that although the average amount in the cans is about 12 ounces (based on sample means), there is a great deal of variability in...
-
Explain how a statistical significant relationship may not be practical for a management decision.
-
In decision theory, payoffs or profits resulting from each possible combination of alternatives and outcomes are called a. conditional values. b. alternatives. c. state of nature. d. none of the...
-
Supertronics, Inc., would like to know how the firms profitability is altered by product mix. Currently, product mix is determined by giving priority to the product with the highest per unit...
-
Beginning with a country that has a trade deficit, demonstrate graphically what will happen to a countrys potential output with globalization if that countrys costs of production fall. Explain your...
-
FIFO method (continuation of 17-30). Do Problem 17-30 using the FIFO method of process costing. Explain any difference between the cost per equivalent unit in the Assembly Department under the...
-
Transferred-in costs, weighted average method (related to 17-30 to 17-32). Larsen Company, as you know, is a manufacturer of car seats. Each car seat passes through the Assembly Department and...
-
Transferred-in costs, FIFO method (continuation of 17-33). Refer to the information in Problem 17-33. Suppose that Larsen Company uses the FIFO method instead of the weighted-average method in all of...
-
3. Many well-operated activated sludge systems have a small layer of light brown foam covering up to 30% of the aeration tank. However, excessive foaming is often an indication of operational...
-
Assets =1 Liabilities + Stockho Accounts Cash + Receivable + Supplies + Equipment Accounts Payable Common + Stock + Revenues Expens 2 +$16,000 1. +$16,000 2. -2,000 +$4,500 +$2,500 3. -650 +$650...
-
Each figure shows an angle with its bisector. 1) m 2 39. Find m/1. 2) Find m/SUT ifm/1-51 U S H F 3) Find m/2 if m/HFG=48. H F 4) Find m/ if m2-18 A

Study smarter with the SolutionInn App


