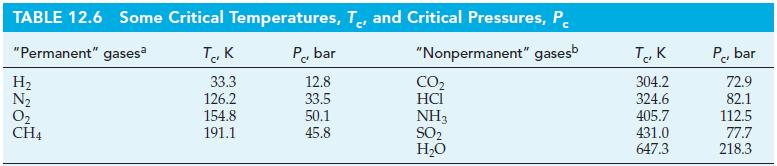
Compare the critical temperatures of NH 3 and N 2 (Table 12.6). Which gas has the stronger
Question:
Compare the critical temperatures of NH3 and N2 (Table 12.6). Which gas has the stronger intermolecular forces?
Table 12.6
Transcribed Image Text:
TABLE 12.6 Some Critical Temperatures, T, and Critical Pressures, P "Permanent" gasesa "Nonpermanent" gasesb H₂ N₂ 0₂ CH4 так 33.3 126.2 154.8 191.1 P, bar 12.8 33.5 50.1 45.8 CO₂ HCI NH3 SO₂ H₂O T, K 304.2 324.6 405.7 431.0 647.3 P, bar 72.9 82.1 112.5 77.7 218.3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
The critical temperature of NH3 4057 K ...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
A vapor mixture having equal volumes of NH3 and N2 is to be contacted at 20?C and 1 atm (760 torr) with water to absorb a portion of the NH3. If 14 m3 of this mixture is brought into contact with 10...
-
3.2 Class Dictionary This class implements a dictionary using a hash table in which collisions are resolved using separate chaining. The hash table will store objects of the class Record. You will...
-
1. How strong are the competitive forces confronting J. Crew in the market for specialty retail? Do a [Michael Porter] five-forces analysis to support your answer. (see chapter 3 in the textfor...
-
T-bills currently yield 3.9 percent. Stock in Nina Manufacturing is currently selling for $63 per share. There is no possibility that the stock will be worth less than $61 per share in one year. a....
-
Benny and Sheree entered into a contract for the sale of one hundred barrels of flour. No mention was made of any place of delivery. Thereafter, Sheree demanded that Benny deliver the flour at her...
-
Propose structures for ketones or aldehydes that have the following 1 H NMR spectra: (a) C 10 H 12 O ? ??IR: 1710 cm ?1 ? (b) C 6 H 12 O 3 ? ? ?IR: 1715 cm ?1 ? (c) C 4 H 6 O ? ? ? ?IR: 1690 cm ?1 ?...
-
In 2015, the city of San Francisco enacted an ordinance that required health warnings on advertisements for certain sugar-sweetened beverages (SSBs) that read: WARNING: Drinking beverages with added...
-
1. Create an initial ERD for the new system that contains at least eight entities. 2. Analyze each relationship to determine if it is 1:1, 1:M, or M:N. 3. Normalize your designs for all tables to...
-
What does SQL stand for? What is SQL? What is SQL used for? What is PL/SQL? What is NoSQL? What is Data Definition Language (DDL)? What is Data Manipulation Language (DML)? What is Data Control...
-
(A) If the reaction described in this example resulted in H 2 O produced and maintained at would the water be present as vapor only or as liquid and vapor in equilibrium? Explain. (B) For the...
-
Arrange the liquids represented by the following molecular models in the expected order of increasing viscosity at 25 C. (a) (b) (c) (d)
-
Evaluate the potential value of incorporating a mobile text-to-win promotion into an offline campaign.
-
Locate several classified ads from the web and/or the Sunday help wanted ads and find one of your choosing. - Analyze the effectiveness of one of these ads you have picked; you only need to analyze...
-
In terms of international management, How can a student with no international experience work on global mind-set, and why is it important to do so? provide supporting details and info backing in...
-
What are some examples of a gap analysis of a non profit organization
-
What does Southwest Airlines need to change in order to facilitate innovation?
-
Q: Detail a market research for a business plan of a small bakery located in Hawthorn, Melbourne. - Market research [ What statistical research have you completed to help you analyse your market?...
-
On January 1, 2013, NRC Credit Corporation leased equipment to Brand Services under a direct financing lease designed to earn NRC a 12% rate of return for providing long-term financing. The lease...
-
As indicated by mutual fund flows, investors tend to beat the market seek safety invest in last year's winner invest in last years loser
-
Calculating Break-Even in each of the following cases, calculate the accounting break-even and the cash break-even points. Ignore any tax effects in calculating the cash break-even. Unit Price Unit...
-
Calculating Break-Even In each of the following cases, find the unknown variable: Accounting Break-Even 127.500 135,000 5,478 Fixed Costs Unit Variable Cost Unit Price Depreciation $30 $41 3.200,000...
-
Calculating Break-Even A project has the following estimated data: price = $68 per unit; variable costs = $41 per unit; fixed costs = $8,000; required return = 15 percent; initial investment =...
-
Let f = C(R) and h > 0. Find constants A, B, C such that f'(x) = A(x + 2h) + Bf(x + h) + Cf(x) + O(h). 2h
-
Let f C (R) and If L = = lim f(h). h0 L = f(h) + ch + C10h 10 + C15h 15 +.. find a linear combination of f(h) and f(h/2) to provide a higher order approxi- mation to L
-
Given the function f, evaluate f (-1), (0), (2), and (4). f(-1) = = = f(0) = f(2) = (4) Number Number = Number = Number x-2 if x < 2 f(x)= 4+|x6| if x 2

Study smarter with the SolutionInn App


