Determine the values of K p from the K c values given. (a) NO4(g) K (b) 2
Question:
Determine the values of Kp from the Kc values given.
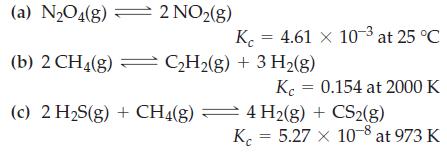
Transcribed Image Text:
(a) N₂O4(g) K (b) 2 CH4(g) = C₂H₂(g) + 3 H₂(g) (c) 2 H₂S(g) + CH4(g) 2 NO₂(g) = 4.61 x 10-3 at 25 °C Kc = 0.154 at 2000 K 4 H2(g) + CS2(g) K = 5.27 x 10-8 at 973 K
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
Here are the conversion factors between Kp and Kc for the given reactions Reaction Kp Kc Conversion ...View the full answer

Answered By

Arun kumar
made more than four thousand assignments
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
You have been assigned the task of measuring the equilibrium constant for the reaction N 2 O 4 2NO 2 as a function of temperature. To do so, you evacuate a rigid 2-liter vessel equipped with a...
-
Consider the nanofluid of Example 2.2. Example 2.2 (a) Calculate the Prandtl numbers of the base fluid and nanofluid, using information provided in the example problem. (b) For a geometry of fixed...
-
A 0.831-g sample of SO3 is placed in a 1.00-L container and heated to 1100 K. The SO3 decomposes to SO2 and O2: At equilibrium the total pressure in the container is 1.300 atm. Find the values of Kp...
-
Because Natalie has been so successful operating Cookie Creations, Katy would like to have Natalie become her partner. Katy believes that together they will create a thriving cookie-making business....
-
Ogden Corporation has compiled the following information on a capital expenditure proposal: (1) The projected cash inflows are normally distributed with a mean of $36,000 and a standard deviation of...
-
In 1967, a Chicago brewer, Meister Brau, Inc., began making and selling a reduced-calorie, reduced-carbohydrate beer under the name LITE. Late in 1968, that company filed applications to register...
-
Pick a product like Cadillac or Sara Lee Deli products or service like Mr. Clean Performance Car Wash or a car and home insurance company that is offered in a limited number of countries. Assess the...
-
Trendy Toes, Co., produces sports socks. The company has fixed costs of $95,000 and variable costs of $0.95 per package. Each package sells for $1.90. Requirements 1. Compute the contribution margin...
-
Define and comprehend the concept of data base and its types. 7. Briefly discuss Database Management Systems. 8. Enumerate and briefly discuss the key concepts of Database Management Systems....
-
(A) Equilibrium is established in a 3.00 L flask at 1405 K for the reaction 2 H 2 S(g) 2 H 2 (g) + S 2 (g). At equilibrium, there is 0.11 mol S 2 (g), 0.22 mol H 2 (g), and 2.78 mol H 2 S(g). What...
-
(A) The reaction N 2 O 4 (g) 2 NO 2 (g) has r H = +57.2 kJ mol -1 . Will the amount of NO 2 (g) formed from N 2 O 4 (g) be greater at high or low temperatures? (B) The enthalpy of formation of NH 3...
-
How can an organization balance its responsibilities to both shareholders and stakeholders?
-
A sleeping 68 kg man has a metabolic power of 73 W . How many Calories does he burn during an 8.0 hour sleep?
-
Far out in space, a mass m=119200.0 kg rocket and a mass m= 221800.0 kg rocket are docked at opposite ends of a motionless 76.0m long connecting tunnel. The tunnel is rigid and has a mass of...
-
A radioactive source consists of a mixture of two radioactive nuclides whose initial activities are identical .One nuclide decays with a half-life of 1/2 year , the other with a half-life of 1/3...
-
In the emptiness of space, or a near vacuum, could you detect, in the absence of instruments, if you were falling under the influence of gravity alone? Explain your answer?
-
Blue corporation has a deficit in accumulated E& P of 400,000 and has current E&P of $360,000. On july 1, Blue distributes $200,000 to its sole shareholder, Sam who has a basis in his stock of...
-
The table below shows the weekly relationship between output and number of workers for a factory with a fixed size of plant. a. Calculate the marginal product of labor b. At what point do diminishing...
-
You are maintaining a subsidiary ledger account for Police-Training Expenditures for 2013. The following columns are used: Inventory purchases are initially recorded as expenditures. Record the...
-
Alcoa Inc. is the worlds largest producer of aluminum products. One product that Alcoa manufactures is aluminum sheet products for the aerospace industry. The entire output of the Smelting Department...
-
Domino Foods, Inc., manufactures a sugar product by a continuous process, involving three production departmentsRefining, Sifting, and Packing. Assume that records indicate that direct materials,...
-
The chief cost accountant for Mountain Glade Beverage Co. estimated that total factory overhead cost for the Blending Department for the coming fiscal year beginning March 1 would be $546,000, and...
-
QUESTION 3 A ball is kicked with an initial velocity of 61 m/s. If the ball's maximum height reached is 71 m, what was the angle of projection? Use -10 m/ss for acceleration. Leave answer to zero...
-
ANSWER EVERYTHING IN DETAIL!! PROVIDE APA REFERENCES!!! 1. Explain IN DETAIL why, from an utility/usefulness standpoint, the following two are interesting and impressive: a) theHarvester b) TCP and...
-
Solving for the unknown resistance Rx, we obtain R (8) R = R3. R

Study smarter with the SolutionInn App


