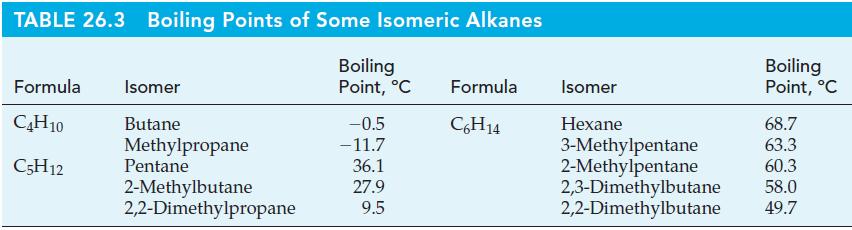
Draw structural formulas for all the isomers listed in Table 26.3, and show that, indeed, the substances
Question:
Draw structural formulas for all the isomers listed in Table 26.3, and show that, indeed, the substances with more compact structures have lower boiling points.
Table 26.3
Transcribed Image Text:
TABLE 26.3 Boiling Points of Some Isomeric Alkanes Boiling Point, °C Formula Isomer C4H10 Butane C5H12 Methylpropane Pentane 2-Methylbutane 2,2-Dimethylpropane -0.5 -11.7 36.1 27.9 9.5 Formula C6H14 Isomer Hexane Boiling Point, °C 68.7 3-Methylpentane 63.3 60.3 2-Methylpentane 2,3-Dimethylbutane 58.0 2,2-Dimethylbutane 49.7
Step by Step Answer:

This question has not been answered yet.
You can Ask your question!
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Draw structural formulas for all possible isomers having the following molecular formulas: a. C3H8 b. C3H7F c. C2H2Br2 d. C3H6 e. C4H9Cl f. C3H6Cl2 g. C3H8S h. C2H4F2
-
Draw structural formulas for the five isomers of C6H14. As you write them out, try to be systematic, starting with a consecutive chain of six carbon atoms.
-
Draw structural formulas for all possible dimethylcyclobutanes. Include cis-trans isomers.
-
Evaluate the limit of the sequence or state that it does not exist. an || u8 n!
-
Jordan Industries is a manufacturing company that produces solid oak office furniture. During the year, the following costs were incurred. The building depreciation and the utilities are allocated...
-
What is the significance of the par value on a preferred stock? What is the significance of the par value on a common stock?
-
The John Gore Organization owns and operates the Charles Playhouse, a theater in Boston, Massachusetts. Evelyn Castillo has diabetes, a disability under the Americans with Disabilities Act (ADA)....
-
Bartlett Company, headquartered in Cincinnati, Ohio, has occasional transactions with companies in a foreign country whose currency is the lira. Prepare journal entries for the following transactions...
-
Assets Cash Accounts receivable Inventory Plant and equipment, net Joel de Paris, Incorporated Balance Sheet Beginning Balance $ 135,000 342,000 Ending Balance $ 135,000 485,000 Investment in...
-
Supply condensed or structural formulas for the following substances. (a) Cycloocta-1,5-diene (an intermediate in the manufacture of resins) (b) 3,7,11-trimethyl-2,6,10-dodecatriene-1-ol...
-
Draw as many structural isomers as you can for cyclic alcohols having the formula C 4 H 6 O. Try to draw at least five. (There are more than five.)
-
Prove each statement for positive integers n and r, with r n. C(0, 0) = 1
-
Calculate standard Enthalpy, Entropy and Gibbs free energy of chemical reaction at 298K using data given to you. Ca(a)+BaO(s)=>CaO(s) +Ba(a) at 298K b) Does this reaction spontaneously take place?...
-
6- A spherical object with mass= 40 g and diameter d= 1.6 cm. Calculate the density of the object in (g/cm) and in (kg/m). Cylinder 1 P (g/cm) P (kg/m)
-
3. Consider a circular loop of wire of radius R in the cy-plane carrying a constant current I going anticlockwise (i.e., in the , direction), in the presence of a uniform magnetic field B = B,. 1 (a)...
-
4. A spaceship is traveling at speed u relative to Earth. The moment the ship passes the Earth, a clock on Earth and a clock on the spaceship both start ticking. After time T (Earth time), the clock...
-
10. Determine the angle a below. 30 a 17 15 11. Determine the value of the sides x and y. Also, determine the value of the angle B. Y B 8 LO
-
Explain the doctrine of respondeat superior.
-
Consider the combustion of methanol below. If 64 grams of methanol reacts with 160 grams of oxygen, what is the CHANGE in volume at STP. 2CH3OH(g) + 3O2(g) 2CO2(g) + 4H2O(1) The volume decreases by...
-
What is operating capital, and why is it important?
-
Explain the difference between NOPAT and net income. Which is a better measure of the performance of a companys operations?
-
What is free cash flow? Why is it the most important measure of cash flow?
-
What is assumption of a mortgage; is it permitted by a typical mortgage instrument; and what rights does the lender have with respect to assumption of the mortgage? How does an assumption of a...
-
Emma Johnson, a 28-year-old software engineer, is purchasing her first home at 123 Sunshine Lane in your state. Emma has been saving for several years and has finally found the perfect single-family...
-
1. How does employment protection legislation actually protect full-time and part-time employees? 2. Do you think that the employment protection legislation provides for effective and reasonable...

Study smarter with the SolutionInn App


