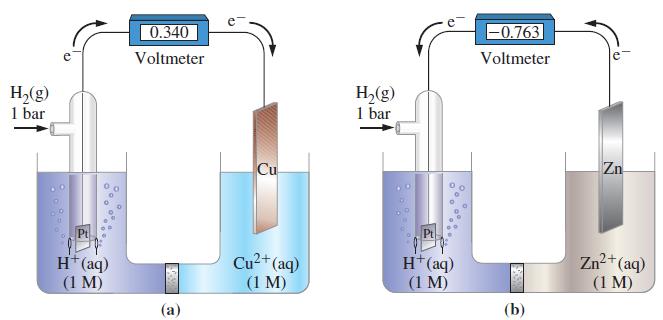
For Figure 19-6, describe any changes in mass that might be detected at the Pt, Cu, and
Question:
For Figure 19-6, describe any changes in mass that might be detected at the Pt, Cu, and Zn electrodes as electric current passes through the electrochemical cells.
Figure 19-6
Transcribed Image Text:
H₂(g) 1 bar Pt H+ (aq) (1 M) 0.340 Voltmeter (a) Cu Cu²+ (aq) (1 M) H₂(g) 1 bar H+ (aq) (1 M) -0.763 Voltmeter (b) Zn Zn²+ (aq) (1 M)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
In Figure 196 we have two different electrochemical cells both containing the same electrolyte solut...View the full answer

Answered By

Asd fgh
sadasmdna,smdna,smdna,msdn,masdn,masnd,masnd,m asd.as,dmas,dma.,sd as.dmas.,dma.,s ma.,sdm.,as mda.,smd.,asmd.,asmd.,asmd.,asm
5.00+
1+ Reviews
15+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
As shown in Fig.P1.41m, air is contained in a vertical piston-cylinder assembly fitted with an electrical resistor. The atmosphere exerts a pressure of 14.lbf/in2 on the top piston, which has a mass...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
An electric cable of radius r l and thermal conductivity k e is enclosed by an insulating sleeve whose outer surface is of radius r 2 and experiences convection heat transfer and radiation exchange...
-
Write a formula for a function g whose graph is similar to f(x) but satisfies the given conditions. Do not simplify the formula. f(x) = 3x 3x + 2 (a) Shifted right 2000 units and upward 70 units (b)...
-
Refer to CVS Corporations annual report in the Supplement to Chapter 5 to answer the following questions. 1. Consolidated balance sheets: a. Did the amount of working capital increase or decrease...
-
Mystic Inc. uses a job order costing system and applies overhead to jobs at a predetermined rate of $4.25 per direct labor dollar. During April 2010, the company spent $29,600 on direct material and...
-
On June 14, 1988, Thomas John Heck Jr. executed a note promising to pay Paul D. Heck \($51,000\) at 7 percent interest compounded annually. The note contains the following payment terms: Perpetual 90...
-
Pietarsaari Oy, a Finnish company, produces cross-country ski poles that it sells for 32 a pair. (The Finnish unit of currency, the euro, is denoted by .) Operating at capacity, the company can...
-
Let ACR be closed and BCR" be compact. Show that the sum set C = A + B = {cR": = a+b for some A, B} is closed. Is it necessarily true that C is compact?
-
(A) The cell diagram for an electrochemical cell is written as Write the equations for the half-cell reactions that occur at the electrodes. Balance the overall cell reaction. (B) The cell diagram...
-
The cell diagram for an electrochemical cell is written as Write the equations for the half-cell reactions that occur at the electrodes. Balance the overall cell reaction. Ni(s)...
-
For u = {1, 2, 3.......9, 10} let A = {1, 2, 3, 4, 5}, B = {1, 2, 4, 8}, C = {1, 2, 3, 5, 7}, and D = {2, 4, 6, 8}. Determine each of the following: (a) (A U B) © C (b) A ª (B © C) (c)...
-
You own a lawn mowing company and are planning to buy some equipment for your company. The equipment will cost you $200,000. From the new equipment you expect to make $70,000 in the first year. Then...
-
5.0 g of aniline (C6H5NH2) is injected into an empty 6.5 L vessel at 140 C. After a long time, how much aniline (in grams) remains in the liquid state? Data: at 140 C, the vapour pressure of aniline...
-
explain the Griffin's logic in using the name Federal Reserve System and appearances of the fourth kind.
-
Draw a timeline for an investment of $40,000 today that returns nothing in one year, $20,000 at the end of year 2, nothing in year 3, and $40,000 at the end of year 4
-
As a process engineer, your task is to design a debutanizer column operating at 1 atm, equipped with a partial reboiler and a total condenser. The feed consists of 900 kg/h of propane, 180 kg/h of...
-
Bill Smith is the Worthington Township manager. When citizens request a traffic light, the staff assesses the traffic flow at the requested intersection. Township policy requires the installation of...
-
Cobb Manufacturing Company uses a process cost system and average costing. The following production data is for the month of June 2011. Production Costs Work in process, beginning of the month:...
-
DeWeese Corporation issues $400,000 of 8%, 5-year bonds on January 1, 2010, at 105. Assuming that the straight-line method is used to amortize the premium, what is the total amount of interest...
-
Mareska Inc. is considering two alternatives to finance its construction of a new $2 million plant. (a) Issuance of 200,000 shares of common stock at the market price of $10 per share. (b) Issuance...
-
Pruitt Corporation issued 3,000, 8%, 5-year, $1,000 bonds dated January 1, 2010, at 100. (a) Prepare the journal entry to record the sale of these bonds on January 1, 2010. (b) Prepare the journal...
-
Two infinitely long wires carry currents 0.81 A and 3.52 A in the same direction. The wires are separated by 29.4 cm. What is the force per unit length that the wires exert on each other? Express...
-
1. A paper ball is flicked off a 1.5-m tall table at a 20 angle from the horizontal with a speed of 5.0 m/s. a. What is the x and y-component of the initial velocity? b. What is the x and y-component...
-
Q.4: Four capacitors are arranged in the following configuration: the capacitances of capacitors are C = 4.0F, C2= 3.0F, C3= 6.0F and C4-2.0F.be 0.0.0.0 (a) Calculate the equivalent capacitance of...

Study smarter with the SolutionInn App


