In one type of Breathalyzer (alcohol meter), the quantity of ethanol in a sample is related to
Question:
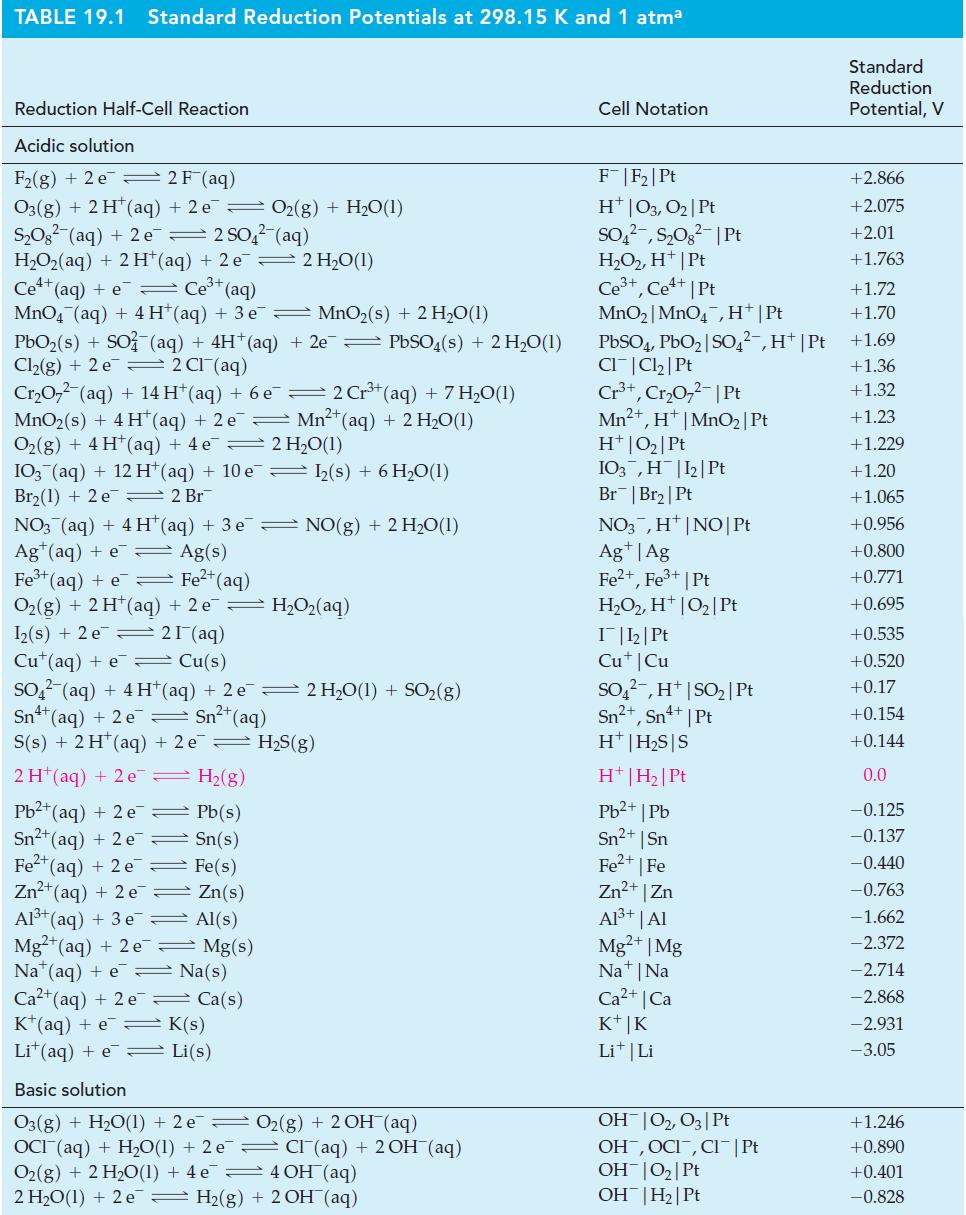
In one type of Breathalyzer (alcohol meter), the quantity of ethanol in a sample is related to the amount of electric current produced by an ethanol–oxygen fuel cell. Use data from Table 19.1 and Appendix D to determine
(a) E°cell and
(b) E° for the reduction of CO2(g) to CH3CH2OH(g).
Table 19.1
Transcribed Image Text:
TABLE 19.1 Standard Reduction Potentials at 298.15 K and 1 atma
Reduction Half-Cell Reaction
Acidic solution
F₂(g) + 2 e 2F (aq)
O3(g) + 2 H¹ (aq) + 2 e O₂(g) + H₂O(1)
S₂O² (aq) + 2 e
2 SO4²(aq)
H,Oz(aq) + 2H*(aq) +2e <2H,O(1)
Ce+ (aq) + e Ce³+ (aq)
MnO4 (aq) + 4H*(aq) + 3 e¯ ⇒ MnO₂(s) + 2 H₂O(1)
PbO₂ (s) + SO² (aq) + 4H+ (aq) + 2e¯ — PbSO(s) + 2 H₂O(1)
Cl₂(g) + 2 e 2 CI (aq)
Cr₂O7² (aq) + 14 H*(aq) + 6 e¯ — 2 Cr³+ (aq) + 7 H₂O(1)
MnO2 (s) + 4H* (aq) + 2 e Mn²+ (aq) + 2 H₂O(1)
O₂(g) + 4H+ (aq) + 4e
2 H₂O(1)
IO3 (aq) + 12 H*(aq) + 10 e
Br₂(1) 2 e 2 Br
NO3(aq) + 4H(aq) + 3 e¯¯ — NO(g) + 2 H₂O(1)
Ag¹(aq) + e
Ag(s)
Fe³+ (aq) + e
Fe²+ (aq)
Oz(g) +2H*(aq) +2e
TABLE 19.1 Standard Reduction Potentials at 298.15 K and 1 atma
Reduction Half-Cell Reaction
Acidic solution
F₂(g) + 2 e 2F (aq)
O3(g) + 2 H¹ (aq) + 2 e O₂(g) + H₂O(1)
S₂O² (aq) + 2 e
2 SO4²(aq)
H,Oz(aq) + 2H*(aq) +2e <2H,O(1)
Ce+ (aq) + e Ce³+ (aq)
MnO4 (aq) + 4H*(aq) + 3 e¯ ⇒ MnO₂(s) + 2 H₂O(1)
PbO₂ (s) + SO² (aq) + 4H+ (aq) + 2e¯ — PbSO(s) + 2 H₂O(1)
Cl₂(g) + 2 e 2 CI (aq)
Cr₂O7² (aq) + 14 H*(aq) + 6 e¯ — 2 Cr³+ (aq) + 7 H₂O(1)
MnO2 (s) + 4H* (aq) + 2 e Mn²+ (aq) + 2 H₂O(1)
O₂(g) + 4H+ (aq) + 4e
2 H₂O(1)
IO3 (aq) + 12 H*(aq) + 10 e
Br₂(1) 2 e 2 Br
NO3(aq) + 4H(aq) + 3 e¯¯ — NO(g) + 2 H₂O(1)
Ag¹(aq) + e
Ag(s)
Fe³+ (aq) + e
Fe²+ (aq)
Oz(g) +2H*(aq) +2e
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
A social worker notices that some of her most anxious clients often come to see her with coffee mugs in hand. Wondering if their anxiousness (X) is related to their amount of caffeine consumption...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-6. On December 12, Irene purchased the building where her store is located. She paid...
-
In the light of what you know about IHRM, how easy do you think it is to manage a global employer brand? What issues would an IHRM professional expect to have to manage? To answer this, analyse the...
-
Rianne Company produces a light fixture with the following unit cost: Direct materials ...... $2 Direct labor ......... 1 Variable overhead ..... 3 Fixed overhead ....... 2 Unit cost ......... $8 The...
-
1. Identify the takeover tactics employed by Mittal. Explain why each one was used. 2. Identify the takeover defenses employed by Arcelor. Explain why each was used. 3. Using the information in this...
-
Identify the most appropriate approach and then design. Provide a rationale as to why this approach and design would be most appropriate. Develop a hypothetical research scenario that would...
-
The following are misstatements that might be found in the client's year-end cash balance (assume that the balance sheet date is June 30): 1. The outstanding checks on the June 30 bank reconciliation...
-
Daniela built a raised garden bed that is 15 feet long, 6 feet wide, and 2.5 feet tall. She plans to fill the garden bed with premium soil that costs $15.96 per cubic yard. Recall that 1 yard = 3...
-
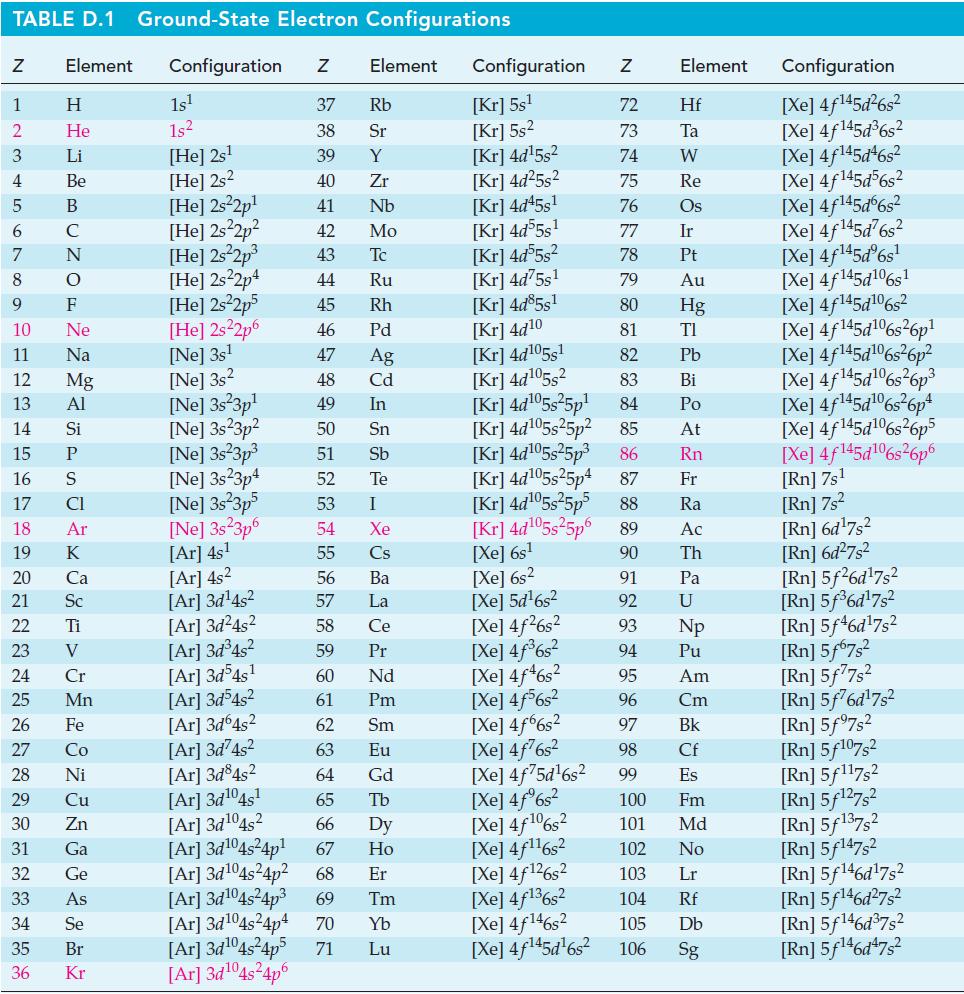
Show that for some fuel cells the efficiency value, e = r G/ r H,can have a value greater than 1.00. Can you identify one such reaction? Use data from Appendix d. TABLE D.1 Ground-State Electron...
-
Isobutylene (2-methylpropene) can be polymerized by treating it with liquid HF as shown in Fig. P5.53. A small amount of tert-butyl fluoride is formed in the reaction. Suggest a curved-arrow...
-
Mr. Keyes was injured on April 30, 2010, when he fell off Ms. Thibodeaux's roof. Mr. Keyes was cleaning and measuring the roof in preparation for painting when, unbeknownst to him, Ms. Thibodeaux...
-
G protein-coupled receptors (GPCRs) and receptor tyrosine kinases (RTKs) are two basic receptor types by which cells transduce signals. Classify the phrases by whether they describe GPCRS, RTKs, both...
-
Compute factory overhead cost from the following costs: Depreciation on factory buildings Depreciation on office equipment Direct materials used Indirect labor $ 45,950 32,980 96,840 $ 6,580
-
Investigate the role of CRISPR-Cas9 technology in genome editing, assessing its potential applications in genetic engineering and disease treatment.
-
Complete the following Common Size Income Statement: Amount Sales $14,000 Cost of Goods Sold 9,000 Percent (b) (c) Gross Profit 5,000 (d) Operating Expenses 2,000 (e) Net Income (a) (f)
-
A motorcycle traveling at 60 m/s accelerates at a rate of 7.0 m/s for 6.0 seconds. What is the final speed of the motorcycle?
-
Hines Moving Company held a fixed-rate debt of $2 million. The company wanted to hedge its fair value exposure with an interest rate swap. However, the only notional available at the time, on the...
-
6 (a) Briefly develop a mathematical model of the behaviour of a copper-twisted pair cable (b) Derive the magnetic energy from: w given that: K + w, where the - - k symbols have their usual meaning...
-
Sam Farr is the president, founder, and majority owner of Galena Medical Corporation, an emerging medical technology products company. Galena is in dire need of additional capital to keep operating...
-
Numerous articles have been written that identify early warning signs that you might be getting into trouble with your personal debt load. You can find many good articles on this topic on the Web....
-
Nordham Corporations trial balance at December 31, 2010, is presented below. All 2010 transactions have been recorded except for the items described below and on the next page. Unrecorded...
-
Jon has an individual account and an UGMA account that he has opened up for his daughter. Both accounts are at the same brokerage house. what is the combined maximum SIPC coverage for both accounts?
-
Suppose in 2015, DBP sold merchandise for $3,500,000 and, based on past experience, recorded a provision for sales returns of $75,000. In 2016, customers returned $50,000 worth of these goods sold to...
-
Has Gap Co acquired another company? If so, what was the acquisition premium (the difference between the price per share offered and the market price per share just before the deal was announced;...

Study smarter with the SolutionInn App


